 FACULTY/GROUP NEWS
FACULTY/GROUP NEWS

Danishefsky Group
Campos Group
Sames Group
 DANISHEFSKY GROUP DANISHEFSKY GROUP
 Researchers in the group of Centennial Professor of Chemistry, Samuel Danishefsky, have synthesized what is arguably the largest and most complex biological molecule ever assembled by the methods of organic chemistry. The molecule is a version of erythropoietin (EPO), a protein critical to the production of red blood cells in mammals and consisting of a folded chain of 166 amino acids with carbohydrates attached at well-defined sites along the chain. Researchers in the group of Centennial Professor of Chemistry, Samuel Danishefsky, have synthesized what is arguably the largest and most complex biological molecule ever assembled by the methods of organic chemistry. The molecule is a version of erythropoietin (EPO), a protein critical to the production of red blood cells in mammals and consisting of a folded chain of 166 amino acids with carbohydrates attached at well-defined sites along the chain.
Professor Danishefsky’s group includes researchers at Columbia and in the Molecular Pharmacology and Chemistry Program at Sloan-Kettering Institute, where Professor Danishefsky is the incumbent of a Eugene W. Kettering Chair. His group’s work on this project was assisted by enabling advances made by Dr. Xianping Tan of Professor Virginia Cornish’s group at Columbia and by Dr. Gong Chen in the Columbia group of Professor Dalibor Sames.
Naturally occurring erythropoietin is actually a highly complex family of molecules, all of which have the same basic chain structure and which differ only in the nature of the attached carbohydrates. The Danishefsky synthesis produces the exact amino acid chain with truncated forms of the carbohydrates attached at the correct locations and with the correct bonding mechanisms. In vitro experiments demonstrate that the synthesized EPO is capable of stimulating the production of red blood cells.
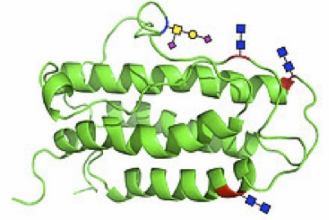 The Danishefsky effort represents the first time a homogenous form of native EPO containing all of the carbohydrates has been obtained. The ability to create a unique variant of EPO, rather than the mix of EPO structures that occur naturally, is a breakthrough that points to eventual progress at systematically isolating, testing, and understanding the behavior of each variant. It also provides a path to address a general question that has potential therapeutic significance -- namely, why does nature produce mixtures of proteins with a unique, underlying structure but which vary only in the nature of a small number of attached carbohydrates? The Danishefsky effort represents the first time a homogenous form of native EPO containing all of the carbohydrates has been obtained. The ability to create a unique variant of EPO, rather than the mix of EPO structures that occur naturally, is a breakthrough that points to eventual progress at systematically isolating, testing, and understanding the behavior of each variant. It also provides a path to address a general question that has potential therapeutic significance -- namely, why does nature produce mixtures of proteins with a unique, underlying structure but which vary only in the nature of a small number of attached carbohydrates?
The new synthesis, which was originally reported online in Angewandte Chemie (1), has subsequently been covered in Science (2), the Royal Society of Chemistry's Chemistry World (3), and C & E News (4) and it will also be reported in Nature. The work is recognized as a breakthrough not only for its success at synthesizing EPO but also for the new techniques developed by the Danishefsky group for fabricating the segments of the EPO structure, for linking them together, and for bonding the carbohydrates at the correct locations. The work represents an effort by the group that started in 2002.
(1) Angew. Chem. Int. Ed. 51, 1 -10, 2012
(2) Science 338, 28, Oct. 2012
(3) RSC Chemistry World, Oct. 10, 2012
(4) C & E News 90, 41, 11, Oct 8, 2012
 CAMPOS GROUP CAMPOS GROUP

On a sunny day in December, twenty-two sixth-grade students from Tech International Charter School in the Bronx came to experience life as a chemistry graduate student. Their afternoon started with a campus tour, with highlights including the old insane asylum, tales of underground railroads, and finally finding the alma mater owl! For a taste of what classes are like, the students were taken to Havemeyer 309, where four graduate students – Emma Dell, Nevette Bailey, Helen Tran, and Miguel Jimenez -- gave presentations to explain their journeys to graduate school. Additionally, the presentation emphasized financial aid and how people can take different paths through education.
The graduate students gave a glimpse into their research, and although an initial answer to the question "What do you think you would get if you combined yeast with the ability to change color and smell?" was "pizza", the students seemed enthused by this project along with stories of stem cells, solar power, and magnets.
The kids then went to the empty lab space on the 13th floor of the North West Corner where the views stretched nearly all the way to their school in the Bronx! Fitted out in lab coat, gloves, and goggles, the students squealed at demonstrations of chemiluminescence and combustion and color changes (see figure). Split into five teams, each named after a Nobel Prize winning scientist, the students then got to try out experiments. First, they prepared their own photographic print by utilizing precipitation reactions and silver chemistry. Then, neutralization chemistry was employed to create a dry ice rainbow and the students investigated how concentration could affect the speed of this reaction.
The day was a huge success! We have set the foundation for a long-standing partnership with the school. As one student wrote in her thank you letter (describing the experiment part of the day) "when my class did this I felt like I was a part of something good". Thanks to Jay Kirschenbaum, Joseph Ulichny, Michael Gaschler, Jose Ricardo Moreno and Chris Torsitano for their help in making this day run so smoothly. The next event with TI Charter will take place on March 20, 2013.
The event was hosted by the Campos Research Group. You can read more about the activities of the Campos Group on their group website.
 SAMES GROUP SAMES GROUP
Stefan Pastine (Ph.D 2006 with Professor Dalibor Sames) recently founded Connora Technologies (www.connoratech.com), a chemistry and materials startup located in Hayward, CA. Connora invented the world’s first recyclable epoxy technology and is poised to change the way in which the composite industry views recycling. Connora’s Recyclamine® epoxy hardener technology enables any epoxy resin system to be recyclable, thus enabling composite manufactures to design fully recyclable fiber-reinforced thermosetting plastics.
Niko Gubernator (Ph.D 2008 with Professor Sames) started a new company called eMolecules, which has been highlighted as the 2nd fastest growing company in San Diego by the San Diego Business Journal. eMolecules has built a powerful chemical search engine combined with customized online ordering and integrated logistical controls. The system allows drug-discovery researchers to source quality compounds from around the globe. eMolecules press releases emphasize that the new system is intended to streamline the ordering process, thereby giving scientists more time to focus on their research objectives. You can read more about the activities of the Sames Group on their group website.
|

