Exam II Chemistry G8248x
Monday, December 12, 1994
Instructions: 50 minutes. Closed book exam.
All material to be graded must appear in your blue book clearly numbered corresponding to the number of the question you are answering. Show your reasoning for full or partial credit.
1. (20 points). The
13C CIDNP spectrum obtained when 99% carbonyl labeled dibenzyl ketone is photolyzed in an NMR spectrometer whose field strength is 10,000 gauss consists of a polarized spectrum corresponding to dibenzyl ketone (carbonyl carbon) at 208 ppm and carbon monoxide at 186 ppm. There is no observable spectrum when the photolyzing lamp is turned off, but when the lamp is turned on again the two peaks reappear. One of the peaks is in absorption and the other is in emission. The two peaks are of equal and opposite intensity. Predict which peak is in enhanced absorption and which is in emission based on (1) the following magnetic parameters for the C6H5CH2CO radical (g = 2.001) and the C6H5CH2 radical (g = 2.003) and the hyperfine coupling constant of +125 gauss and (2) the standard paradigms for photochemistry of ketones.
2. (20 points). Interpret, in terms of a state correlation diagram, the observation that the photolysis of cyclobutanone occurs efficiently via
a-cleavage even at very low temperatures, whereas cyclohexanone is relative stable to a-cleavage at the same temperature.
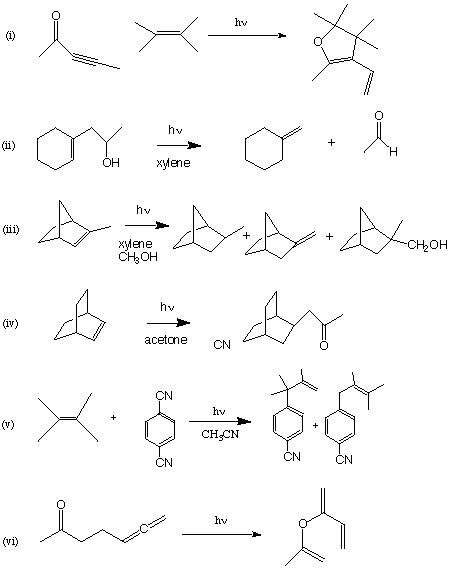
3. (60 points. 10 Points Each Question). Suggest plausible mechanisms for the following overall photochemical reactions. In some cases the indicated products may be products of secondary photolysis of another photoproduct.
