

Xenotransplantation:E T H I C S I N R E S E A R C H
huge benefits, hard choices
Organ transplants save lives, but the supply is short. Transplants of animal organs
might be an answer.
SOMETIME SOONŁŁprobably this fall, if he gets approval from
the Institutional Review Board at Columbia- What
Michler wants to do raises issues that he himself calls "mystical." Animal tissues have been
used to benefit humans for years, but implanting an entire organ seems different.
Michler's ideal candidate will be within days of death and of an appropriate size, too small for
a bulky left-ventricular assist device ("mechanical heart"). Both patient and family will
be capable of making decisions. The patient will have exhausted the medical and surgical
options, but his or her other organs will be sound. "Probably someone on the waiting list for a
heart transplant," Dr. Michler says. "I see this first transplant as a bridge, buying time of
weeks to months until a human heart is found."
Next step in surgical history?
"Use of animal cells or tissue could be beneficial in AIDS, Parkinson's disease,
diabetes, spinal cord injury, acute liver failure, psoriasis, muscular dystrophy,
myocardial infarction, and other
conditions," says Norman G. Levinsky, chairman of Boston University's department of
medicine, speaking at a June conference on xenografting sponsored by the Institute of Medicine and the National Academy of Sciences (IOM/NAS).
Michler adds: "As a cardiac surgeon, I'm faced with people who die all the time. It's abhorrent.
My interest in xenografts is a direct response to a tremendous problem, the management of
heart failureŁŁthe No. 1 cause of death in this country today. At least 45,000 of those people
could be saved by a heart transplant, but we can transplant only about 2,000 because of the
shortage of donor organs."
It may not be possible to increase the number of human organs available. "Presumed consent"
laws in Belgium and Austria mean that potential donors are assumed to have authorized
transplantation unless they have opted out. But in his country, many argue, such a law might
be perceived as exploitation of minorities and the poor. "People may feel the physician is not
doing everything to save the person's life because he wants to use the person as a donor,"
Michler says. In expanding the number of available organs, asserts Penn sociologist Renee Fox,
transplanters are "beginning to slide down a slippery slope."
There's another slippery slope here, besides the dehumanizing of the dying process. Unless we
use animal grafts there will be abuses: organs are already being sold
"There's another slippery slope here, besides the dehumanizing of the dying process," counters
David Rothman, professor of social medicine and director of the Center for Study of
Society and Medicine at P&S. "Animal grafts may prevent other abuses. For example,
China and Taiwan use organs from executed prisoners; in India, it's commonplace to let
people sell their organs."
Starzl has called for a moratorium on xenotransplants as permanent replacements; his team
performed two, and both patients died from immunologic problems. But xenografting can
extend a candidate's life while a human donor is sought. CPMC has been a leading
transplantation center for years. Its heart-transplant service, first under the direction of
Keith Reemtsma, professor and former chairman of surgery, and later under Michler and
Eric Rose, chairman of surgery, is the largest in the nation. Thirty years ago, at Tulane University, Reemtsma transplanted several
chimpanzee kidneys to
humans. The longest survivor, a woman of 23, lived for nine months. Columbia faculty
helped launch the recent IOM/NAS meeting on xenografting and raised the crucial issue of
possible transmission of infectious diseases. Ralph Dell, professor of pediatrics and head of the
medical center's Institutional Animal Care and Use Committee, called researchers at the Centers for Disease Control and Prevention, the NIH, and the FDA. A
national meeting to consider social, ethical, and legal issues of xenotransplantation was being
discussed, but infectious diseasesŁŁ"the most critical issue," Dell saysŁŁwere not on the agenda
until he persuaded the IOM and NAS to add these issues to the program. The
IOM/NAS meeting in June was followed by a meeting of the FDA. After public comment,
final guidelines will be issued. Says Dell: "The proposed guidelines say that animal sources
should be screened for various viral diseases and not used if certain ones are found. Early
research in xenotransplantation should be done at medical centers with expertise in
transplantation and experience in virology and immunology. That means large academic
medical centers such as Columbia-Presbyterian, Stanford, and Pittsburgh." "There are different
types of xenograftsŁŁbetween close species or between distant species, and whole-organ
transplants or transplants of free tissues such as pancreatic islet cells, bone marrow, or skin,"
says Jeffrey Platt, professor of experimental surgery at Duke.
Whole-organ transplantsŁŁwhether xenografts or allograftsŁŁmay have immunologic
effects on the lining of blood vessels, leading to immediate or chronic rejection. Free tissues or
cells cause fewer rejection problems. Introducing human genes into the animal donor to make
its organs more compatible might also reduce rejection. Continued
Pigs are easier to raise and have organs resembling ours. More than 90 million pigs are
slaughtered each year in the United States for food, and many of their heart valves are used to
repair human hearts after a tanning process renders the valves immunologically inert. Because
pigs are genetically further from humans, pig organs are more likely to cause rejection but less
likely to transmit viruses. To lessen rejection, Michler and others are breeding transgenic pigs,
which contain parts of the human immune system.
Veterinarian Jonathan Allan of the Southwest Foundation
for Biomedical Research, a primate
center, riveted the IOM/NAS conference when he said, "Don't do it. Using baboon tissue in
humans is dangerous."
Although baboons and pigs can be raised in "specific pathogen-free" colonies, that
means only that they do not have the pathogens we can test for. "How can you screen for
something when you don't know it's there?" Dr. Allan asked. "HIV is the best example. How
long can you quarantine patients to protect against transmission of a cancer virus that might
take 10 or 20 years to show itself? Up to 50 percent of African monkeys carry an
AIDS-like virus [simian immunodeficiency virus, or SIV]. It's no problem, no disease,
for them"ŁŁyet HIV-1 and HIV-2 are widely believed to have evolved from their
close relative SIV because of monkey-human blood contact. "Every single
retrovirus has the possibility of causing cancer," Allan warns. Some cause only minor disease
in the monkey but are lethal in humans. "AIDS is the best example of what we don't want to
do. We need new, reliable assays for baboon viruses," Allan said. Pigs, too, harbor retroviruses.
"The idea that we're going to create a plague is defused by the hundreds of thousands of
transplant patients who've returned to their pets, farms, homes, and families," Starzl said.
They have not spread viruses, despite receiving immunosuppressive drugs. Furthermore,
human exposure to blood does not seem to have led to major outbreaks of disease, with the
possible exception of AIDS. In Africa, some monkeys are hunted. Farmers and butchers
everywhere are exposed to animal blood, but no major outbreaks of disease have been
identified among them.
"The concern is with viruses, like the AIDS virus or hepatitis B, that are present in blood and
body fluids. If it were a respiratory virus, we'd already have it," says Dell. Universal
precautions to avoid contact with body fluids will protect health care workers. Recipients of
animal organs would be advised to practice safe sex, avoid donating blood, and undergo
careful follow-up by physicians.
"Scientists alternate between 'what if' and 'so what,'" says Stephen Morse of Rockefeller
University, an authority on emerging viruses.
He points out that a new epidemic would require two steps: viral infection of a person and
transmission of the virus to others. Before xenotransplantation is done, he says, monitoring
systems should be set up to seek unusual events in recipients, health care workers, and family
members. Further risks and rewards
Like allografting and other
costly procedures, xenografting may defy economic valuation; the cheapest choice, after all,
would be death. "The cost is irrelevant," asserts Michler. "We're in the business of saving lives.
I deal with a patient on a one-to-one basis." "This is an ethical subject
created by the relationship of humans to animals," Starzl says. "All of society ought to decide.
Regulating something that doesn't exist yet is the best way to ensure that it won't exist."
Do the potentially life-saving benefits of xenotransplantation outweigh the risks and
costs? Michler's answer is an unqualified yes. He quotes ethicist M.R. Shimkin: "To do
nothing, or to prevent others from doing anything, is itself a type of experiment, for the
prevention of experimentation is tantamount to the assumption of responsibility for an
experiment different from the one proposed."(2) The
FDA agrees. Despite reservations about the chance of success, an advisory panel in July gave
the go-ahead for one bone-marrow transplant from a baboon to a human with
AIDS. By the time this article is read, the transplant may already have taken place at the University of California, San Francisco.
LAST YEAR, researchers at Columbia-Presbyterian
sparked a nationwide debate about xenotransplantation. When Dr. Robert Michler submitted
his protocol to the CPMC Institutional Review Board (IRB) and Institutional Animal Care
Utilization Committee in April 1994 to perform a baboon-to-human heart
transplant, the committees decided three areas needed further examination: the current status
of immunologic research, infectious disease issues, and ethical issues. The discussions at
a June 1994 seminar satisfied committee members that all the pertinent issues had been
adequately covered except one: the potential for an epidemic of transspecies infection. The
committees determined that this was outside the scope of their mandates, since one exists to
ensure humane care of animals in experiments and the other looks out for individual patients'
interests. According to Ralph Dell, "neither group is constituted to look after the public
health."
Thus, the Institute of Medicine, the Food and Drug Administration, the Centers for Disease
Control and Prevention, the National Institutes of Health, and the Department of Defense all
have considered the immunologic, technical, ethical, and public policy implications of
xenotransplantation.
The IOM/NAS meeting concluded that xenotransplants should proceed and institutions need
to monitor themselves. On July 13 and 14, 1995, the FDA Biological Response Modifiers
Advisory Committee met to decide whether to recommend that the FDA allow a baboon
bone-marrow transplantation.
Even researchers who support xenotransplantation express concern about pathogen
transmission. Researchers are not as concerned about highly infective viruses that manifest
quickly (such as Ebola) as they are about retroviruses, which can exist in the host for a long
period before becoming apparent. Non-human primates being considered as donors are
known to carry herpesviruses and retroviruses. The simian agents' ability to cause disease in
humans is poorly defined.
At a joint meeting of CPMC's IRB and animal-care committee on July 26, a discussion
focused on a type of retrovirus called spumaviruses or foamy viruses. While foamy viruses are
not endemic to newborn baboons, 80 to 100 percent acquire them from other baboons in the
colony. It is unknown whether the foamy virus causes disease in humans, but it has been
associated with neurodegenerative disease in mice. One solution is to exclude carrier baboons;
however, its prevalence would make this difficult.
The FDA committee ultimately decided to recommend approval of the marrow graft,
reasoning that proposed precautions will minimize the risk of spreading an unknown
pathogen. The committee also decided not to prohibit any baboon transplants but to leave
responsibility for approval up to IRBs and animal-care committees. Meanwhile,
guidelines are soon to be published in the Federal Register.
Dr. Michler and his colleagues have been asked to provide a revised protocol, including viral
surveillance procedures for baboon donors, exclusion criteria, and monitoring methods for
patients. The CPMC committees will then decide whether to proceed. At press time, no
decision had been made.
Transspecies transplants (xenografts) are being considered today because of the dramatic
success of human-to-human transplants, or allografts (see illustration). "More
than 100,000 people are alive today with successfully transplanted human organs," says
transplant pioneer Thomas Starzl of the University of
Pittsburgh. More than 40,000
Americans are on waiting lists for human organs; one-third to one-half will die
before an organ can be found.


1628: Attempts at blood
transfusion,
a form of transplantation, by Giovanni Colle; deaths lead to banning of the
practice
1901: Karl Landsteiner discovers ABO blood types, allowing safer transfusion
1950: First kidney allograft performed by Richard Lawler
1963: First liver allograft performed by Thomas Starzl; first lung allograft by James
Hardy; mechanical cardiac assist device implanted by Michael DeBakey
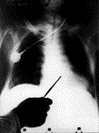
1967: First human heart transplanted by Christiaan Barnard in Cape Town, South
Africa. Chest film shows location of heart in recipient Louis Washkansky

1972: Cyclosporin, powerful immunosuppressant that minimizes tissue rejection,
developed at Sandoz Laboratories
1992: Starzl transplants baboon liver into human patient

Any day now: Baboon-to-human heart xenograft
CREDITS: 1) BETTMANN ARCHIVE; 2) GUYDOSH/SYGMA; 3) AP/WIDE
WORLD PHOTOS;
Choosing a species
Chimpanzees are humans' closest genetic kin, sharing more than 99 percent of our genome,
minimizing the chance of rejection. But they are an endangered species. That leaves two
choices: baboons and pigs. Baboons are closely related to humans, lessening rejection risks but
increasing our unease at exploiting a cousin-like speciesŁŁand raising the risk of
transmitting human pathogens.
Other ethical considerations include the use of animals and of financial resources in an era of
managed care and tight research funding. Animal-rights activists strongly oppose
xenografting, although they are less vocal about porcine insulin, heart valves, and skin grafts.
Some religious groups might object to xenotransplantation as "playing God."(1) The costs of xenotransplantation also raise questions of
access: "Who pays for it? Who gets it? Somebody has hypertension and can't get the medicine
because of lack of insurance, so he ends up in the hospital and gets a baboon heart," says Dr.
Stuart Youngner of University Hospitals in Cleveland.
Will there be monkey business
at CMPC?
Related links...
JANICE
HOPKINS TANNE is a contributing editor for New York and
an award-winning medical journalist whose work has appeared in the British
Medical Journal, American Health, GQ, Mirabella, and the New York Times
Health Supplement. She recently served as president of the American Society of Journalists and Authors.




