| ||
For a copy of these pages in pdf format, click here. I. Research While a Student
1. Thomas J. Katz and John L. Margrave, "Free Energy Functions for Gaseous Atoms from Hydrogen (Z = 1) to Niobium (Z = 41)," J. Chem. Phys. 1955, 23, 983. 
II. Ten-Pi-Electron AromaticsThe cyclooctatetraene dianion (1), the cyclooctatetraene anion-radical (2), the cyclononatetraenyl anion (3), and the pentalene dianion (4) were synthesized and characterized. Ions 1 and 3 were the first analogues of benzene to be prepared in which ten pi-electrons, rather than the six found in benzene, are delocalized in a ring, and 4 was the first isolable derivative of pentalene. Related structures were also prepared. Noteworthy are the experiments that showed 1 and the corresponding anion-radical (2) to be planar, unlike cyclooctatetraene itself. Noteworthy too is a novel procedure used to synthesize 3. In addition, the experiments turned up an effect that iodide ions exert on the course of carbene-additions to olefins. The availability of ions 1–3 helped to determine the relationships between carbon charge density and 13C and 1H NMR chemical shifts and between carbon radical density and proton hyperfine splittings. 1. Thomas J. Katz, "The Cycloöctatetraenyl Dianion," J. Am. Chem. Soc. 1960, 82, 3784–3785. 2. Thomas J. Katz, "The Chemical Interaction of the Cyclooctatetraenyl Anions," J. Am. Chem. Soc. 1960, 82, 3785–3786. 3. Thomas J. Katz and Herbert L. Strauss, "ESR Spectrum of the Cyclooctatetraenyl Radical Anion," J. Chem. Phys. 1960, 32, 1873–1875. 4. Thomas J. Katz, William H. Reinmuth, and Donald E. Smith, "Electrolytic Reduction of Cycloöctatetraene," J. Am. Chem. Soc. 1962, 84, 802–808. 5. Herbert L. Strauss, Thomas J. Katz, and George K. Fraenkel, "Electron Spin Resonance Studies of the Cyclooctatetraenyl Anions," J. Am. Chem. Soc. 1963, 85, 2360–2364. 6. Thomas J. Katz and Peter J. Garratt, "The Cyclononatetraenyl Anion," J. Am. Chem. Soc. 1963, 85, 2852. 7. Thomas J. Katz and Peter J. Garratt, "Reactions of the Cyclooctatetraenyl Dianion with gem-Dihalides. The Preparation of Derivatives of Bicyclo[6.1.0]nonatriene. Synthesis of the Cyclononatetraenyl Anion," J. Am. Chem. Soc. 1964, 86, 5194–5202. 8. Thomas J. Katz and Michael Rosenberger, "The Pentalenyl Dianion," J. Am. Chem. Soc. 1962, 84, 865–866. 9. Thomas J. Katz, Michael Rosenberger, and Raymond K. O'Hara, "The Pentalenyl Dianion," J. Am. Chem. Soc. 1964, 86, 249–252. 10. W. H. Okamura and T. J. Katz, "The 4-Azapentalenyl Anion," Tetrahedron 1967, 23, 2941–2957. 11. Thomas J. Katz, Masayuki Yoshida, and L. C. Siew, "The sym-Dibenzcyclooctatetraene Anion Radical and Dianion," J. Am. Chem. Soc. 1965, 87, 4516–4520. 12. Thomas J. Katz and Wojciech Ślusarek, "The Trindene Trianion," J. Am. Chem. Soc. 1980, 102, 1058–1063. 13. Nancy Acton, Donald Hou, Joshua Schwarz, and Thomas J. Katz, "Preparation and Oxidation of the Bis(tetra-n-butylammonium) Salt of 2,2'-(2,7-Pyrenediyl)bis[propane- dinitrile] Dianion," J. Org. Chem. 1982, 47, 1011–1018. Back to Top 
III. Isotope Effects and the Mechanism of CycloadditionIt was shown for the first time how a secondary deuterium isotope effect could be used to analyze the mechanism of a cycloaddition reaction. As an adjunct, the experiments turned up a rearrangement that cyclobutanones undergo when exposed to acid.1. Thomas J. Katz and Ralph Dessau, "Hydrogen Isotope Effects and the Mechanism of Cycloaddition," J. Am. Chem. Soc. 1963, 85, 2172–2173. Back to Top 
IV. Homoaromatic IonsCation 5 and related structures were the first cyclobutenyl cations synthesized. Their ultraviolet spectra, which exhibit absorption maxima at wavelengths between those of simple allylic and cyclopropenium cations, measured the effect of cross-ring resonance. An analysis of the electron spin resonance spectrum showed the structure of another homoaromatic ion, 6. 1. Thomas J. Katz, J. Richard Hall, and William C. Neikam, "1,2,3,4-Tetramethyl- 2-chlorocyclobutenyl Cation," J. Am. Chem. Soc. 1962, 84, 3199–3200. 2. Thomas J. Katz and Elijah H. Gold, "The Synthesis and Properties of Cyclobutenyl Cations," J. Am. Chem. Soc. 1964, 86, 1600–1606. 3. Elijah H. Gold and Thomas J. Katz, "Preparation of the 4-Iodo-1,2,3,4-tetra- methylcyclobutenyl Cation. Ultraviolet and Nuclear Magnetic Resonance Spectra of Cyclobutenyl Cations in Methylene Chloride, Sulfuric Acid, and Liquid Sulfur Dioxide," J. Org. Chem. 1966, 31, 372–374. 4. Thomas J. Katz and Carolyn Talcott, "The Cyclononatetraene Anion Radical," J. Am. Chem. Soc. 1966, 88, 4732–4733. 5. William H. Okamura, William H. Snider, and Thomas J. Katz, "The Synthesis of Homoazepine Derivatives," Tetrahedron Lett. 1968, 3367–3370. Back to Top 
V. Applications of Aromatic Anions in Organic SynthesisIons 1 and 3 (see section II) were used to make other ring systems such as 7–11. Structure 8 was the first well-characterized phosphirane. (An earlier patent by Wagner claimed the preparation of the parent phosphirane and recorded two of its characteristics, its melting point and its vapor pressure.) The thermal transformations of 8 into 9, of 9 into its stereoisomer, and of 10 into 11 were analyzed.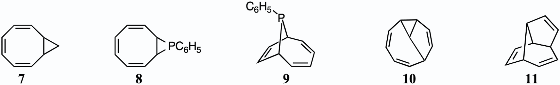 1. Thomas J. Katz and Peter J. Garratt, "The Reaction of the Cyclooctatetraenyl Dianion with gem-Dihalides. The Addition of Alkyl Carbenes to Cyclooctatetraene," J. Am. Chem. Soc. 1964, 86, 4876–4879. 2. Thomas J. Katz, Christina R. Nicholson, and C. A. Reilly, "The Reaction of Dipotassium Cyclooctatetraenide with Dichlorophenylphosphine. Rearrangements of 9-Phenyl-9-phosphabicyclo[6.1.0]nonatriene and 9-Phenyl-9-phosphabicyclo[4.2.1]- nonatriene," J. Am. Chem. Soc. 1966, 88, 3832–3842. 3. Thomas J. Katz, James C. Carnahan, Jr., George M. Clarke, and Nancy Acton, "The Photochemical Synthesis of Condensed Bridged Phosphines," J. Am. Chem. Soc. 1970, 92, 734–735. 4. Thomas J. Katz and J. J. Cheung, "A Simple Synthesis of a C10H10 Hydrocarbon and an Explanation of C10H10 Photochemistry," J. Am. Chem. Soc. 1969, 91, 7772–7774. 5. Thomas J. Katz, J. J. Cheung, and Nancy Acton, "The Synthesis of Isobullvalene," J. Am. Chem. Soc. 1970, 92, 6643–6644. Back to Top 
VI. Metal-Sandwich CompoundsThe first molecules were synthesized in which two metal atoms are sandwiched between a pair of planar aromatic rings (12 and related metallocenes), as were the first ansa-titanocene, 13, and related structures. Combined with ethylaluminumdichloride, 13 polymerizes ethylene.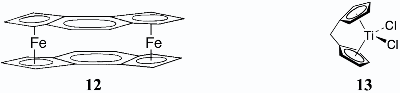 1. Thomas J. Katz and Jerome Schulman, "The as-Indacenyl Dianion and Bis(as- indacenyliron)," J. Am. Chem. Soc. 1964, 86, 3169–3170. 2. Thomas J. Katz, Valeria Balogh, and Jerome Schulman, "Synthesis of Bis(as- indacenyliron)," J. Am. Chem. Soc. 1968, 90, 734–739. 3. Ramsey Gitany, Iain C. Paul, Nancy Acton, and Thomas J. Katz, "The Structure of Bis(as-indacenyliron)," Tetrahedron Lett. 1970, 2723–2726. 4. Thomas J. Katz and Joseph J. Mrowca, "The Pentalenylcycloocta-1,5-dienerhodium Anion and Hydropentalenyl Complexes of Thallium, Platinum, and Rhodium," J. Am. Chem. Soc. 1967, 89, 1105–1111. 5. Thomas J. Katz and Nancy Acton, "Bis(pentalenylnickel)," J. Am. Chem. Soc. 1972, 94, 3281–3283. 6. Thomas J. Katz, Nancy Acton, and James McGinnis, "Sandwiches of Iron and Cobalt with Pentalene," J. Am. Chem. Soc. 1972, 94, 6205–6206. 7. Thomas J. Katz and Michael Rosenberger, "Ferrocene Derivatives of Pentalene: Dipentalenyliron Dianion and Hydrodipentalenyliron Anion," J. Am. Chem. Soc. 1963, 85, 2030. 8. Thomas J. Katz, Nancy Acton, and Geoffrey Martin, "The [1n]Ferrocenophanes," J. Am. Chem. Soc. 1969, 91, 2804. 9. Thomas J. Katz, Nancy Acton, and Geoffrey Martin, "Preparation of Metallocenes from Hydrocarbon Dianions," J. Am. Chem. Soc. 1973, 95, 2934–2939. 10. Thomas J. Katz and Wojciech Ślusarek, "Helical Hydrocarbons Capped by Five-Membered Rings as Precursors of Polymeric Metallocenes," J. Am. Chem. Soc. 1979, 101, 4259–4267. 11. Thomas J. Katz and Nancy Acton, "1,1'-Methylenetitanocene Dichloride," Tetrahedron Lett. 1970, 2497–2499. 12. Monika E. Huttenloch, Josef Diebold, Ursula Rief, Hans H. Brintzinger, Adam M. Gilbert, and Thomas J. Katz, "Biphenyl-Bridged Metallocenes That Are Chiral, Configurationally Stable, and Free of Diastereomers," Organometallics 1992, 11, 3600-3607. 13. Thomas J. Katz, Adam M. Gilbert, Monika E. Huttenloch, Gu Min-Min, and Hans H. Brintzinger, "Palladium-Catalyzed Coupling of 2,2'-Dihalobiaryls with Metallated Cyclopentenes. An Easy Synthesis for Spiro[Cyclopentene-1,9'-[9H]Fluorene]s," Tetrahedron Lett. 1993, 34, 3551–3554. Back to Top 
VII. Rhodium-Catalyzed Cycloaddition ReactionsThe papers below report the discovery that rhodium catalyzes cycloaddition reactions, Eqs. 1–3 and related transformations.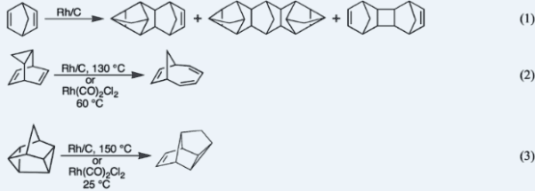 1. Joseph J. Mrowca and Thomas J. Katz, "Catalysis of a Cycloaddition Reaction by Rhodium on Carbon," J. Am. Chem. Soc. 1966, 88, 4012–4015. 2. Thomas J. Katz, James C. Carnahan, Jr., and Roy Boecke, "The Preparation of Tetracyclo[4.3.0.02,4.03,7]non-8-ene and the Dimerization of It and of Benzo- norbornadiene by Rhodium on Carbon," J. Org. Chem. 1967, 32, 1301–1304. 3. Thomas J. Katz, Nancy Acton, and Iain C. Paul, "The Reaction of Complexes of Rhodium(I) Chloride with Norbornadiene," J. Am. Chem. Soc. 1969, 91, 206–208. 4. Thomas J. Katz and Steven Cerefice, "Thermodynamics of the Interconversion of Certain Bridged Ring Hydrocarbons by Rhodium Catalysts," Tetrahedron Lett. 1969, 2509–2510. 5. Thomas J. Katz and Steven A. Cerefice, "Rhodium Catalyzed Isomerization of Tricyclo[3.2.2.02,4]nonadiene," Tetrahedron Lett. 1969, 2561–2563. 6. Nancy Acton, Ronald J. Roth, Thomas J. Katz, JoAnn K. Frank, Carol A. Maier, and Iain C. Paul, "Dimerization and Trimerization of Norbornadiene by Soluble Rhodium Catalysts," J. Am. Chem. Soc. 1972, 94, 5446–5456. Back to Top 
VIII. Mechanism of Metal-Catalyzed Cycloaddition ReactionsThe mechanism by which rhodium catalyzes cycloaddition reactions was indicated by analyses that identified (a) the stereochemical course of the rhodium-catalyzed dimerization in Eq. 1 (section VII) and (b) the structure of a reduced dimer formed when the dimerization is carried out in the presence of hydrogen. The mechanism was shown definitively for the first time by experiments that proposed and applied a new technique, subsequently named by Carpenter the induced kinetic isotope effect. The key experimental observation is that when the transformation in equation 4 below is carried out to only a small extent with a mixture of 14 and the analogue of 14 that has no deuterium atoms, deuterium accumulates greatly in the electrocyclic reaction product, 15, even though the formation of 15 entails no carbon-hydrogen bond-breaking. The implication is that the pathways to all three products must branch from one irreversibly formed precursor, and since the precursor of the last two products (16 and 17) must be a metallacycle (18 or a valence-tautomer of 18), that same metallacycle must be the precursor of the first (15). This was the first demonstration that metallacycles are intermediates in metal-catalyzed electrocyclic transformations.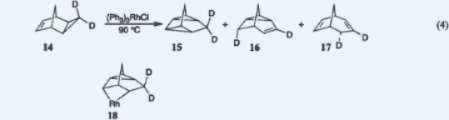 1. Thomas J. Katz and Nancy Acton, "The Stereochemical Course of Metal Catalyzed Cycloaddition Reactions of Norbornadiene," Tetrahedron Lett.1967, 2601–2605. 2. F. Peter Boer, Melvin A. Neuman, Ronald J. Roth, and Thomas J. Katz, "The Structure of Binor-S Established from the Dione Derivative. The Crystal and Molecular Structure of Decahydro[1,2,4:5,6,8]dimetheno-s-indacenedione," J. Am. Chem. Soc.1971, 93, 4436–4442. 3. Ronald J. Roth and Thomas J. Katz, "The Hydrogenation of Norbornadiene," Tetrahedron Lett.1972, 2503–2504. 4. Thomas J. Katz and Steven A. Cerefice "The Mechanism of a Metal-Catalyzed Cycloaddition Reaction," J. Am. Chem. Soc.1969, 91, 6519–6521. 5. Thomas J. Katz and Steven Cerefice, "The Reaction of exo-Tricyclo[3.2.1.02,4]- octene with Rhodium Catalysts," J. Am. Chem. Soc. 1969, 91, 2405–2406. 6. Thomas J. Katz and Steven A. Cerefice, "The Stereochemistry of a Rhodium-Catalyzed Rearrangement of a Cyclopropane to a Propylene," J. Am. Chem. Soc.1971, 93, 1049–1050. 
IX. Valence Isomers of BenzeneThe three valence isomers of benzene, 19–21, were synthesized. Benzvalene (19), previously obtainable only in minuscule amounts, could be prepared easily in quantity and could then be used to prepare other structures. Prismane (20) was obtained in only very small amounts, but there is no other way to obtain it. A notable, but not obvious, rearrangement was analyzed that occurs when electrophiles add to benzvalene. 1. Thomas J. Katz, E. Jang Wang, and Nancy Acton, "A Benzvalene Synthesis," J. Am. Chem. Soc. 1971, 93, 3782–3783. 2. Thomas J. Katz, Ronald J. Roth, Nancy Acton, and Eileen Jang Carnahan, "Synthesis of Benzvalene," J. Org. Chem. 1999, 64, 7663–7664. 3. Ronald J. Roth and Thomas J. Katz, "Uses of Benzvalene in Synthesis. A Synthesis of Tricyclo[2.2.0.02,6]hexane," J. Am. Chem. Soc. 1972, 94, 4770–4771. 4. Thomas J. Katz and Nancy Acton, "Synthesis of Prismane," J. Am. Chem. Soc. 1973, 95, 2738–2739. 5. Thomas J. Katz and Kyriacos C. Nicolaou, "Additions of Chlorosulfonyl Isocyanate and Sulfenyl Halides to Benzvalene," J. Am. Chem. Soc. 1974, 96, 1948–1949. 6. Ronald J. Roth and Thomas J. Katz, "Reactions and Rearrangements of 5,6-Disubstituted Bicyclo[2.l.l]hexenes," J. Org. Chem. 1980, 45, 96l–965. 7. Nicholas J. Turro, Carl A. Renner, Walter H. Waddell, and Thomas J. Katz, "Thermolysis and Photochemistry of Cyclic Azo Compounds. A Novel Chemiluminescent Reaction and Alternate Route to Dewar Benzene," J. Am. Chem. Soc. 1976, 98, 4320–4322. 8. Nicholas J. Turro, Peter Lechtken, Arthur Lyons, Richard R. Hautala, Eileen Carnahan, and Thomas J. Katz, "Photochemical Generation of Electronically Excited Organic Products in Adiabatic Pericyclic Photoreactions. An Unexpected Propensity toward Spin Inversion in a Retrocycloaddition and in a Valence Isomerization," J. Am. Chem. Soc. 1973, 95, 2035–2037. 9. Carl A. Renner, Thomas J. Katz, Joseph Pouliquen, Nicholas J. Turro, and Walter H. Waddell, "Energy Storage and Release. Direct and Sensitized Photoreactions of Benzvalene. Evidence for a Quantum Chain Process, an Adiabatic Photorearrangement, a Degenerate Photovalence Isomerization, and Two Reactive Triplet States," J. Am. Chem. Soc. 1975, 97, 2568–2570. 10. Nicholas J. Turro, Carl A. Renner, Thomas J. Katz, Kenneth B. Wiberg, and Helen A. Connon, "Kinetics and Thermochemistry of the Rearrangement of Benzvalene to Benzene. An Energy Sufficient but Non-Chemiluminescent Reaction," Tetrahedron Lett. 1976, 4133–4136. 11. Nicholas J. Turro, V. Ramamurthy, and Thomas J. Katz, "Energy Storage and Release. Direct and Sensitized Photoreactions of Dewar Benzene and Prismane," Nouv. J. Chim. 1977, 1, 363–365. 12. Barry M. Trost, Paul H. Scudder, Robert M. Cory, Nicholas J. Turro, V. Ramamurthy, and Thomas J. Katz, "l,2-Diaza-2,4,6,8-cyclooctatetraene," J. Org. Chem. 1979, 44, 1264–1269. Back to Top 
X. Synthesis of PentaalkylphosphoranesThe addition of metal alkyls to phosphonium salts constrained to small rings (illustrated in Eq. 5) was the first and is still the only procedure known to give pentasubstituted alkylphosphorus structures.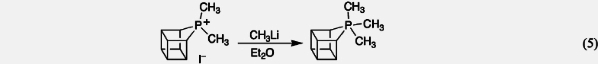 1. E. Wayne Turnblom and Thomas J. Katz, "A Stable Pentaalkylphosphorane," J. Am. Chem. Soc. 1971, 93, 4065–4066. 2. E. Wayne Turnblom and Thomas J. Katz, "Synthesis of Alkylphosphoranes," J. Am. Chem. Soc. 1973, 95, 4292–4311. 3. Thomas J. Katz and E. Wayne Turnblom, "Stable Cycloalkyl Pentavalent Phosphoranes," J. Am. Chem. Soc. 1970, 92, 6701–6702. 4. C. Hackett Bushweller, Howard S. Bilofsky, E. Wayne Turnblom, and Thomas J. Katz, "Barriers to Pseudorotation in Alkylphosphoranes," Tetrahedron Lett. 1972, 2401–2404. 5. E. Wayne Turnblom and Thomas J. Katz, "Angular Dependence of Phosphorane and Ylide Formation," J. Chem. Soc., Chem. Commun. 1972, 1270–1271. 6. James P. Albarella and Thomas J. Katz, "Preparation and Rearrangement of Bridgehead Phosphorus Ylides and Their Derivatives in the Homocubane Ring System," J. Org. Chem. 1978, 43, 4338–4345. Back to Top 
XI. Mechanism of the Olefin Metathesis ReactionThe differences between the mechanistic and experimental analyses in the papers below and in the paper published earlier by Hérisson and Chauvin are not commonly recognized. The paper by Hérisson and Chauvin reported three experiments. In one, a cyclic olefin was combined with a 1,2-disubstituted olefin, and the claim was that initially such reactions should and do give 1:2:1 ratios of products. However, initially the ratios should in fact not be 1:2:1; it is only at equilibrium that this ratio should be obtained. Thermodynamics is independent of mechanism, and the observation of a 1:2:1 ratio therefore has no mechanistic implications. In Hérisson and Chauvin's second experiment, a cyclic olefin was combined with a terminal olefin, and only one product was observed where three were observed in the first experiment. This observation led Hérisson and Chauvin to conclude that "this simple representation [the metal-carbene propagated mechanism for olefin metathesis] does not provide an explanation for … the observed distribution [of products] in the coreaction of cyclic olefins and α-olefins" [emphasis added; original in French]. In their third experiment, Hérisson and Chauvin showed that metathesis with 2-pentene causes 1,5-cyclooctadiene and 1,5,9-cyclododecatriene to fragment, and they reported that this too was irreconcilable with the metal-carbene mechanism.In fact, if metal-carbenes propagate olefin metatheses, all such experiments must give the products in ratios that initially are 1:r:1, where r > 2, and the greater the difference between the two metal-carbenes that derive from the cleavage of a double bond, the larger r should be. In our experiments, the initial ratio of products when a cyclic olefin (cyclooctene) was combined with a 1,2-disubstituted olefin (2-hexene) and a molybdenum catalyst (Mo[(C6H5)3P]2Cl2(NO)2 + (CH3)3Al2Cl3) was not 1:2:1, but 1:3.25:1. This result and the results that Hérisson and Chauvin said do not support the metal-carbene mechanism are the ones that do support that mechanism; the experiment that Hérisson and Chauvin said does support the mechanism is the one that does not. Other experiments and more extensive analyses distinguishing alternative mechanistic proposals are described in the publications of 1975 and 1977 and summarized in the review of 1977. These publications also explain the fragmentation of 1,5-cyclooctadiene and 1,5,9-cyclododecatriene, which Hérisson and Chauvin reported not to be in accord with the metal-carbene-propagated mechanism. The 1975 paper and 1977 review also make a notable point that has been omitted from subsequent discussions of the evidence for the mechanism of the olefin metathesis reaction: the observation that acetylenes metathesize provided one of the strongest pieces of evidence against the then most commonly accepted alternative mechanism. This observation also inspired the proposal that metal-carbynes propagate the metathesis of acetylenes. Also not commonly recognized is the relationship between two sets of publications: (1) the 1975 and 1977 papers and 1977 review below and (2) reference 2 below (from 1976) and similar papers by Grubbs. Both showed that metathesis does not simply interchange the moieties of two alkenes. The former set of papers showed this for reactions in which cyclic alkenes combine with terminal alkenes, the latter set for the same reactions in reverse. But only the former set, by explaining the previously inexplicable distribution of products that arise when cyclic alkenes combine with acyclic alkenes, eliminated contradictory evidence that previously had appeared to exclude the metal-carbene-propagated mechanism, reconciled all other experimental observations with this mechanism, and pointed out the mechanistic significance of the acetylene metathesis. These are the results, along with the demonstration that metal-carbenes initiate the reaction, that established the mechanism of olefin metathesis. 1. Thomas J. Katz and James McGinnis, "The Mechanism of the Olefin Metathesis Reaction," J. Am. Chem. Soc. 1975, 97, 1592–1594. 2. Thomas J. Katz and Robert Rothchild, "Mechanism of the Olefin Metathesis of 2,2'-Divinylbiphenyl," J. Am. Chem. Soc. 1976, 98, 2519–2526. 3. Thomas J. Katz and James McGinnis, "Metathesis of Cyclic and Acyclic Olefins," J. Am. Chem. Soc. 1977, 99, 1903–1912. 4. Thomas J. Katz, "The Olefin Metathesis Reaction," Adv. Organometal. Chem. 1977, 16, 283–317. 5. James McGinnis, Thomas J. Katz, and Samuel Hurwitz, "Selectivity in the Olefin Metathesis of Unsymmetrically Substituted Ethylenes," J. Am. Chem. Soc. 1976, 98, 605–606. Back to Top 
XII. Initiations of Olefin Metatheses and Acetylene Polymerizations by Metal-CarbenesIsolable metal-carbenes were shown to initiate metatheses of olefins and polymerizations of acetylenes. The use of these initiators averted acid-catalyzed side reactions and gave rise to uniquely high stereospecificity. Ten years after the papers below reported the ability of metal-carbenes to initiate olefin metatheses and the benefits these initiators provide, Schrock reported a tungsten-carbene that was much more active and the following year a molybdenum-carbene that was even more useful. Six years after Schrock's report, Grubbs described a ruthenium-carbene that was still more useful for olefin metatheses, though not for acetylene polymerizations. However, the experiments in the papers below were the first in which a metal-carbene initiated olefin metatheses and the first in which a metal-carbene initated polymerizations of acetylenes. The review of 2005 (reference 2 below) shows that vague complaints about the merit of these experiments are themselves without merit.These experiments provided the first examples of metatheses of trisubstituted olefins and showed, in accord with the idea described in section XI, that since one of the two metal-carbenes that might propagate an olefin metathesis should be favored over the other, cyclic trisubstituted olefins give polymers that are translationally invariant. The stereochemistry of olefin metathesis was analyzed for the first time, and metathesis products were produced with high cis-stereoselectivity. Isotope-labeling experiments provided evidence for the mechanisms of acetylene polymerizations initiated by metal-derivatives. A new transformation of thioacetylenes induced by metal-carbenes was also uncovered. 1. Thomas J. Katz, "Fischer Metal Carbenes and Olefin Metathesis," In Handbook of Metathesis; Robert H. Grubbs, Ed.; Wiley-VCH: Weinheim, Germany, 2003; Volume 1, Chapter 5. 2. Thomas J. Katz, "Olefin Metatheses and Related Reactions Initiated by Carbene Derivatives of Metals in Low Oxidation States," Angew. Chem. 2005, 117, 3070–3079; Angew. Chem. Int. Ed. 2005, 44, 3010–3019. 3. Thomas J. Katz, James McGinnis, and Craig Altus, "Metathesis of a Cyclic Trisubstituted Alkene. Preparation of Polyisoprene from 1-Methylcyclobutene," J. Am. Chem. Soc. 1976, 98, 606–608. 4. Steven J. Lee, James McGinnis, and Thomas J. Katz, "Directional Specificity and Stereoselectivity in the Metathesis of a Trisubstituted Olefin," J. Am. Chem. Soc. 1976, 98, 7818–7819. 5. Thomas J. Katz, Steven J. Lee, and Michael A. Shippey, "Preparations of Polymers Using Metal-Carbenes," J. Mol. Catal. 1980, 8, 219–226. 6. Thomas J. Katz, Steven J. Lee, and Nancy Acton, "Stereospecific Polymerizations of Cycloalkenes Induced by a Metal-Carbene," Tetrahedron Lett. 1976, 4247–4250. 7. Thomas J. Katz and Nancy Acton, "Metatheses Induced by (Phenylmethoxy- carbene)pentacarbonyltungsten," Tetrahedron Lett. 1976, 4251–4254. 8. Thomas J. Katz and Steven J. Lee, "Initiation of Acetylene Polymerization by Metal Carbenes," J. Am. Chem. Soc. 1980, 102, 422–424. 9. Thoi Huu Ho and Thomas J. Katz, "Polymerization of Functionalized Acetylenes," J. Mol. Catal. 1985, 28, 359–367. 10. Thomas J. Katz, Thoi Huu Ho, Neng-Yang Shih, Yuan-Chi Ying, and Van I. W. Stuart, "Polymerizations of Acetylenes and Cyclic Olefins Induced by Metal Carbynes," J. Am. Chem. Soc. 1984, 106, 2659–2668. 11. Thomas J. Katz and William H. Hersh, "The Stereochemistry of the Olefin Metathesis Reaction," Tetrahedron Lett. 1977, 585–588. 12. T. C. Clarke, C. S. Yannoni, and T. J. Katz, "Mechanism of Ziegler-Natta Polymerization of Acetylene: A Nutation NMR Study," J. Am. Chem. Soc. 1983, 105, 7787–7789. 13. Thomas J. Katz, Scott M. Hacker, R. D. Kendrick, and C. S. Yannoni, "Mechanisms of Phenylacetylene Polymerization by Molybdenum and Titanium Initiators," J. Am. Chem. Soc. 1985, 107, 2182–2183. 14. Thomas J. Katz, Ginger Xu-Qiang Yang, Barry H. Rickman, and Takashi Iwashita, "Transformation of Alkynyl Thioethers into Disubstituted Acetylenes by Combination with Metal-Carbenes," J. Am. Chem. Soc. 1993, 115, 2038–2039. Back to Top 
XIII. The Enyne Metathesis ReactionThe enyne metathesis reaction (Eq. 6) was brought about for the first time as an outgrowth of the idea that acetylenes should and do induce otherwise inactive metal-derivatives to initiate olefin metatheses. The experiments showed that the pericyclic rearrangements of metallacyclobutenes that take place during these enyne metatheses are highly stereoselective and give (Z)-metallabutadienes (Eq. 7). The transformations were initiated by isolable metal-carbenes, and the mechanism of the initiation was identified by demonstrating that, under appropriate conditions, a fragment of the initiator attaches to the product. Variations of these experiments led to other transformations of enynes and diynes.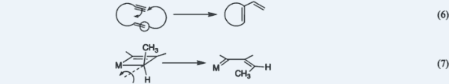 1. Thomas J. Katz and Timothy M. Sivavec, "Metal-Catalyzed Rearrangement of Alkene-Alkynes and the Stereochemistry of Metallacyclobutene Ring Opening," J. Am. Chem. Soc. 1985, 107, 737–738. 2. Thomas J. Katz, "Reactions of Acetylenes and Alkenes Induced by Catalysts of Olefin Metathesis," in Advances in Metal Carbene Chemistry, U. Schubert (editor); Kluwer Academic Publishers; Dordrecht, Netherlands, 1989; pp 293–304. 3. Thomas J. Katz, Steven J. Lee, Mridula Nair, and Edward B. Savage, "Induction of Olefin Metathesis by Acetylenes," J. Am. Chem. Soc. 1980, 102, 7940–7942. 4. Thomas J. Katz, Edward B. Savage, Steven J. Lee, and Mridula Nair, "Reactivities of Metal Carbenes toward Alkenes and Alkynes," J. Am. Chem. Soc. 1980, 102, 7942–7944. 5. Thomas J. Katz and Chien-Chung Han, "Induction of Olefin Metathesis by Phenylacetylene plus Tungsten Hexachloride," Organometallics 1982, 1, 1093–1095. 6. Chien-Chung Han and Thomas J. Katz, "A Procedure Identifying a Polyacetylene Initiator of Olefin Metathesis. The Reactivities of Metal Carbenes toward Alkenes and Alkynes," Organometallics 1985, 4, 2186–2195. 7. Thomas J. Katz and Ginger Xu-Qiang Yang, "Reactions of Enynes with Adsorbed Metal-Carbenes," Tetrahedron Lett. 1991, 32, 5895–5898. 8. Timothy M. Sivavec and Thomas J. Katz, "Synthesis of Phenols from Metal-Carbynes and Diynes," Tetrahedron Lett. 1985, 26, 2159–2162. 9. Timothy M. Sivavec, Thomas J. Katz, Michael Y. Chiang, and Ginger Xu-Qiang Yang, "A Metal Pentadienyl Prepared by Reacting a Metal Carbyne with an Enyne," Organometallics 1989, 8, 1620–1625. Back to Top 
XIV. Synthesis of Helical Conjugated Metallocenes and Helical Conjugated PolymersHelical metallocenes (22) and helical metallocene oligomers (23), all enantiopure, were synthesized by an unprecedented process that uses an asymmetric center in the precursors to control the direction in which the helices wind. Helical metallocenes had previously been unknown. The first helical conjugated polymer to be synthesized is shown in Eq. 8.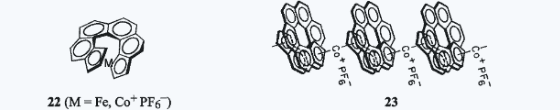 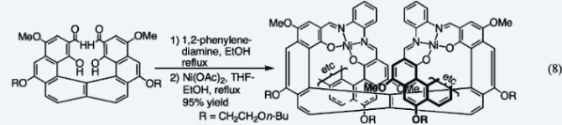 1.Thomas J. Katz and Jaan Pesti, "Synthesis of a Helical Ferrocene," J. Am. Chem. Soc. 1982, 104, 346–347. 2. Anantha Sudhakar and Thomas J. Katz, "Asymmetric Synthesis of Helical Metallocenes," J. Am. Chem. Soc. 1986, 108, 179–181. 3. Thomas J. Katz, Anantha Sudhakar, Bingwei Yang, and James S. Nowick, "Synthesis of Conjugated Optically Active Polymetallocenes," J. Macromol. Sci., Chem. 1989, A26, 309–326. 4. Adam M. Gilbert, Thomas J. Katz, William E. Geiger, Matthew P. Robben, and Arnold L. Rheingold, "The Synthesis and Properties of an Optically Active Helical Bis-Cobaltocenium Ion," J. Am. Chem. Soc. 1993, 115, 3199–3211. 5. Anantha Sudhakar, Thomas J. Katz, and Bing-Wei Yang, "Synthesis of a Helical Metallocene Oligomer," J. Am. Chem. Soc. 1986, 108, 2790–2791. 6. Thomas J. Katz, Anantha Sudhakar, Mark F. Teasley, Adam M. Gilbert, William E. Geiger, Matthew P. Robben, Martin Wuensch, and Michael D. Ward, "The Synthesis and Properties of Optically Active Helical Metallocene Oligomers," J. Am. Chem. Soc. 1993, 115, 3182–3198. 7. Michael K. Poindexter and Thomas J. Katz, "Simple Conversion of Aryl Bromides to Arylmethylphosphonates," Tetrahedron Lett. 1988, 29, 1513–1516. 8. Yujia Dai, Thomas J. Katz, and David A. Nichols, "Synthesis of a Helical Conjugated Ladder Polymer," Angew. Chem. 1996, 108, 2230–2232; Angew. Chem., Int. Ed. Engl. 1996, 35, 2109–2111. 9. Yujia Dai and Thomas J. Katz, "Synthesis of Helical Conjugated Ladder Polymers," J. Org. Chem. 1997, 62, 1274–1285. Back to Top 
XV. Stilbene PhotocyclizationsThe now dominant procedure for effecting stilbene photocyclizations, employing propylene oxide and stoichiometric amounts of iodine, was discovered. So was a procedure (developed in the course of the research in section XIV) that uses bromine substituents to direct the course of photocyclizations.1. Longbin Liu, Bingwei Yang, Thomas J. Katz, and Michael K. Poindexter, "Improved Methodology for Photocyclization Reactions, " J. Org. Chem. 1991, 56, 3769–3775. 2. Anantha Sudhakar and Thomas J. Katz, "Directive Effect of Bromine on Stilbene Photocyclizations. An Improved Synthesis of [7]Helicene," Tetrahedron Lett. 1986, 27, 2231–2234. 3. Bingwei Yang, Longbin Liu, Thomas J. Katz, Charles A. Liberko, and Larry L. Miller, "Electron Delocalization in Helical Quinone Anion-Radicals," J. Am. Chem. Soc. 1991, 113, 8993–8994. 4. Charles A. Liberko, Larry L. Miller, Thomas J. Katz, and Longbin Liu, "The Electronic Structure of Helicene-Bis-Quinone Anion Radicals," J. Am. Chem. Soc. 1993, 115, 2478–2482. 5. Longbin Liu and Thomas J. Katz, "Bromine Auxiliaries in Photosyntheses of [5]Helicenes," Tetrahedron Lett. 1991, 32, 6831–6834. Back to Top 
XVI. Chemistry of Thiazyl DerivativesThe chemistry of thiazyl derivatives was developed to provide simple procedures for aminating allylic positions, for forming thiadiazoles, and for peparing thiadiazyl chlorides, and therefore thiazyl heterocycles. There is an interesting explanation for why the yields are erratic when thionyl chloride transforms carbamates into N-sulfinylcarbamates, and it shows how the problem can be overcome.1. Thomas J. Katz and Shuhao Shi, "A Simple Allylic Amination Procedure and The Metathesis of N-Sulfinylcarbamates," J. Org. Chem. 1994, 59, 8297–8298. 2. Shuhao Shi, Thomas J. Katz, Bingwei V. Yang, and Longbin Liu, "The Use of Thiazyl Chlorides, Alkyl Carbamates, and Thionyl Chloride to Fuse 1,2,5-Thiadiazoles to Quinones and to Oxidize, Chlorinate, and Aminate Them," J. Org. Chem. 1995, 60, 1285–1297. Back to Top 
XVII. Synthesis of Usefully Functionalized Helicenes in QuantityUnlike those available before, the procedure illustrated in Eq. 9 yields helicenes of many different kinds, in amounts that are enormous, and with significant functional groups. Camphanate-derivatives of helicenols were found to be unusually easily resolved into their enantiomers. Particularly interesting is the discovery of why they are so effective.A procedure was developed that uses alcoholic acid to modify alkoxyl substituents on polycyclic aromatic molecules, and it has a significant benefit. It places hydroxyl groups, and through their agency other substituents, specifically into the most sterically hindered positions of helicene structures.  1. Longbin Liu and Thomas J. Katz, "Simple Preparation of a Helical Quinone," Tetrahedron Lett. 1990, 31, 3983–3786. 2. Nikolaos D. Willmore, Longbin Liu, and Thomas J. Katz, "A Diels-Alder Route to [5]- and [6]-Helicenes," Angew. Chem. 1992, 104, 1081; Angew. Chem., Int. Ed. Engl. 1992, 31, 1093. 3. Thomas J. Katz, Longbin Liu, Nikolaos D. Willmore, Joseph M. Fox, Arnold L. Rheingold, Shuhao Shi, Colin Nuckolls, and Barry H. Rickman, "An Efficient Synthesis of Functionalized Helicenes," J. Am. Chem. Soc. 1997, 119, 10054–10063. 4. Joseph M. Fox, Naomi R. Goldberg, and Thomas J. Katz, "An Efficient Synthesis of Functionalized [7]Helicenes," J. Org. Chem. 1998, 63, 7456–7462. 5. Nikolaos D. Willmore, Diego A. Hoic, and Thomas J. Katz, "Diels-Alder Reactions of α-Substituted Styrenes with p-Benzoquinone," J. Org. Chem. 1994, 59, 1889–1891. 6. Tienthong Thongpanchang, Kamil Paruch, Thomas J. Katz, Arnold L. Rheingold, Kin-Chung Lam, and Louise Liable-Sands, "Why (1S)-Camphanates Are Excellent Resolving Agents for Helicen-1-ols and Why They Can Be Used to Analyze Absolute Configurations," J. Org. Chem. 2000, 65, 1850–1856. 7. Kamil Paruch, Thomas J. Katz, Christopher Incarvito, Kin-Chung Lam, Brian Rhatigan, and Arnold L. Rheingold, "First Friedel-Crafts Diacylation of a Phenanthrene as the Basis for an Efficient Synthesis of Nonracemic [7]Helicenes," J. Org. Chem. 2000, 65, 7602–7608. 8. Spencer D. Dreher, Kamil Paruch, and Thomas J. Katz, "Acidified Alcohols as Agents to Introduce and Exchange Alkoxyls on the Periphery of Helicenes," J. Org. Chem. 2000, 65, 806–814. 9. Joseph M. Fox and Thomas J. Katz, "Conversion of a [6]Helicene into an [8]Helicene and a Helical 1,10-Phenanthroline Ligand," J. Org. Chem. 1999, 64, 302–305. 10. Spencer D. Dreher, Daniel J. Weix, and Thomas J. Katz, "An Easy Synthesis of Functionalized Hetero[7]helicenes," J. Org. Chem. 1999, 64, 3671–3678. 11. Karen E. S. Phillips, Thomas J. Katz, Steffen Jockusch, Andrew J. Lovinger, and Nicholas J. Turro, "Synthesis and Properties of an Aggregating Heterocyclic Helicene," J. Am. Chem. Soc. 2001, 123, 11899–11907. 12. Kamil Paruch, Libor Vyklický, and Thomas J. Katz, "Preparation of 9,10-Dimethoxyphenanthrene and 3,6-Diacetyl-9,10-Dimethoxyphenanthrene," Organic Syntheses 2003, 80, 227–232. 13. Kamil Paruch, Libor Vyklický, and Thomas J. Katz, "Helicenebisquinones: Synthesis of a [7]Helicenebisquinone," Organic Syntheses 2003, 80, 233–238. 14. Kamil Paruch, Libor Vyklický, David Zhigang Wang, Thomas J. Katz, Christopher Incarvito, Lev Zakharov, and Arnold L. Rheingold, "Functionalizations of [6]- and [7]Helicenes at Their Most Sterically Hindered Positions," J. Org. Chem. 2003, 68, 8539–8544. 15. Porntip Charoonniyomporn, Tienthong Thongpanchang, Suteera Witayakran, Yodhathai Thebtaranonth, Karen E.S. Phillips, and Thomas J. Katz, "A Convenient One-Pot Synthesis of Bisalkylthioarenes," Tetrahedron Lett. 2004, 45, 457–459. 16. David Zhigang Wang, Thomas J. Katz, James Golen, and Arnold L. Rheingold, "Diels-Alder Additions of Benzynes within Helicene Skeletons," J. Org. Chem. 2004, 69, 7769–7771. Back to Top 
XVIII. Helicenes Useful for Preparative ChemistryHelicene ligands useful for asymmetric catalysis and for the analysis of chirality remote from functional groups were designed and synthesized. Figure 1 illustrates an application.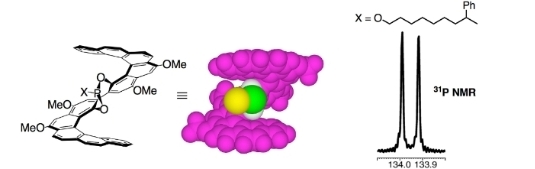 Figure 1. On the left is [5]HELOL chlorophosphite, a reagent that analyzes chirality remote from functional groups. On the right is the 31P NMR spectrum when the reagent is attached to (±)-8-phenylnonanol. It shows the resolution (the scale is ppm) achieved in the analysis of the diastereomers. 1. Spencer D. Dreher, Thomas J. Katz, Kin-Chung Lam, and Arnold L. Rheingold, "Application of the Russig-Laatsch Reaction to Synthesize a Bis[5]Helicene Chiral Pocket for Asymmetric Catalysis," J. Org. Chem. 2000, 65, 815–822. 2. Daniel J. Weix, Spencer D. Dreher, and Thomas J. Katz, "[5]HELOL Phosphite: A Helically Grooved Sensor of Remote Chirality," J. Am. Chem. Soc. 2000, 122, 10027–10032. 3. David Zhigang Wang and Thomas J. Katz, "A [5]HELOL-Analogue that Senses Remote Chirality in Alcohols, Phenols, Amines, and Carboxylic Acids," J. Org. Chem. 2005, 70, 8497–8502. Back to Top 
XIX. Self-Assembly and Optical Properties of Helical Conjugated MoleculesAppropriately substituted helicenes spontaneously aggregate in solution into helical columnar structures. In the absence of solvent, the long corkscrew-shaped columns assemble, as illustrated in Figure 2, into large soft-crystalline fibrous structures, also illustrated in the figure. The materials act as electrooptical switches, fluoresce circularly polarized light, and exhibit a variety of unprecedented properties: enormous specific rotations, high second-order nonlinear optical response, the ability to generate quasi-phase matched second harmonics from unpoled materials, and the ability to reverse optical activity effects not by reversing a sample's chirality but by reversing the direction of an electric field applied to the sample.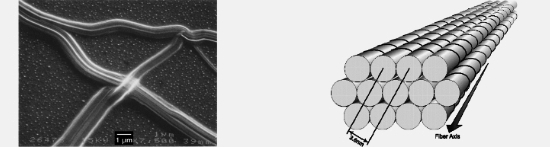 Figure 2. On the left is a scanning electron micrograph of fibers formed by the first helical conjugated molecule that self-aggregates. On the right is a diagram illustrating how columns of the helicenes are organized in the fibers. 1. Colin Nuckolls, Thomas J. Katz, and Louis Castellanos, "Aggregation of Conjugated Helical Molecules," J. Am. Chem. Soc. 1996, 118, 3767–3768. 2. Colin Nuckolls, Thomas J. Katz, Gail Katz, Peter J. Collings, and Louis Castellanos, "Synthesis and Aggregation of a Conjugated Helical Molecule," J. Am. Chem. Soc. 1999, 121, 79–88. 3. Kamil Paruch, Libor Vyklický, Thomas J. Katz, Christopher D. Incarvito, and Arnold L. Rheingold, "Expeditious Procedure to Synthesize Ethers and Esters of Tri- and Tetrahydroxy[6]helicenebisquinones From the Dye-Intermediates Disodium 4-Hydroxy- and 4,5-Dihydroxynaphthalene-2,7-disulfonates," J. Org. Chem. 2000, 65, 8774–8782. 4. Andrew J. Lovinger, Colin Nuckolls, and Thomas J. Katz, "Structure and Morphology of Helicene Fibers," J. Am. Chem. Soc. 1998, 120, 264–268. 5. Colin Nuckolls, Thomas J. Katz, Thierry Verbiest, Sven Van Elshocht, Hans-Georg Kuball, Stefan Kiesewalter, Andrew J. Lovinger, and André Persoons, "Circular Dichroism- and UV-Visible Absorption Spectra of the Langmuir-Blodgett Films of an Aggregating Helicene," J. Am. Chem. Soc. 1998, 120, 8656–8660. 6. Colin Nuckolls and Thomas J. Katz, "Synthesis, Structure, and Properties of a Helical Columnar Liquid Crystal," J. Am. Chem. Soc. 1998, 120, 9541–9544. 7. Libor Vyklický, S. Holger Eichhorn, and Thomas J. Katz, "Helical Discotic Liquid Crystals," Chem. Mater. 2003, 15, 3594–3601. 8. Thierry Verbiest, Sven Van Elshocht, Martti Kauranen, Louis Hellemans, Johan Snauwaert, Colin Nuckolls, Thomas J. Katz, and André Persoons, "Strong Enhancement Of Nonlinear Optical Properties Through Supramolecular Chirality," Science (Washington, DC, U.S.) 1998, 282, 913–915. 9. Thierry Verbiest, Sven Van Elshocht, André Persoons, Colin Nuckolls, Karen E. Phillips, and Thomas J. Katz, "Second-order Nonlinear Optical Properties of Highly Symmetric Chiral Thin Films," Langmuir 2001, 17, 4685–4687. 10. Bertrand Busson, Martti Kauranen, Colin Nuckolls, Thomas J. Katz, and André Persoons, "Quasi phase matching in chiral materials," Phys. Rev. Lett. 2000, 84, 79–82. 11. Thierry Verbiest, Sonja Sioncke, André Persoons, Libor Vyklický, Thomas J. Katz, "Electric Field-Modulated Circular-Difference Effects in Second-Harmonic Generation from a Chiral Liquid Crystal," Angew. Chem. 2002, 114, 4038–4040; Angew. Chem. Int. Ed. 2002, 41, 3882–3884. 12. Joseph M. Fox, Thomas J. Katz, Sven Van Elshocht, Thierry Verbiest, Martti Kauranen, André Persoons, Tienthong Thongpanchang, Todd Krauss, and Louis Brus, "Synthesis, Self-Assembly, and Nonlinear Optical Properties of Conjugated Helical Metal Phthalocyanine Derivatives," J. Am. Chem. Soc. 1999, 121, 3453–3459. 13. Thomas J. Katz, "Syntheses of Functionalized and Aggregating Helical Conjugated Molecules," Angew. Chem. 2000, 112, 1997–1999; Angew. Chem. Int. Ed. 2000, 39, 1921–1923. 14. S. Van Elshocht, B. Busson, T. Verbiest, M. Kauranen, J. Snauwaert, L. Hellemans, A. Persoons, C. Nuckolls, and T. J. Katz, "Enhancement of nonlinear optical properties through supramolecular chirality," Mater. Res. Soc. Symp. Proc. 1999, 561 (Organic Nonlinear Optical Materials and Devices), 15–20. 15. S. Van Elshocht, T. Verbiest, B. Busson, M. Kauranen, J. Snauwaert, L. Hellemans, A. Persoons, C. Nuckolls, K. E. Phillips, and T. J. Katz, "Nonlinear Optical Study of Helicenebisquinones," Synth. Met. 2000, 115, 201–205. 16. S. Van Elshocht, T. Verbiest, T. J. Katz, C. Nuckolls, B. Busson, M. Kauranen, and A. Persoons, "Molecular Chirality as a Tool for Second-order Nonlinear Optics," MCLC S&T, Sect. B: Nonlinear Opt. 2000, 25, 105–115. 17. S. Van Elshocht, T. Verbiest, G. de Schaetzen, L. Hellemans, K. E. S. Phillips, C. Nuckolls, T. J. Katz, and A. Persoons, "Beneficial effect of heating on the morphology and second-order nonlinear optical efficiency of anisotropic thin films," Chem. Phys. Lett. 2000, 323, 340–344. 18. Sonja Sioncke, Sven Van Elshocht. Thierry Verbiest, André Persoons, Martti Kauranen, Karen E. S. Phillips, Thomas J. Katz, "Optical activity effects in SHG from anisotropic chiral thin films," J. Chem. Phys. 2000, 113, 7578–7581. 19. Martti Kauranen, Bertrand Busson, Sven Van Elshocht, Thierry Verbiest, André Persoons, Colin Nuckolls, and Thomas J. Katz, "Second-Order Nonlinear Optics Based on Chiral Materials," Optics and Photonics News 2000, 11 (12), 24–25. 20. S. Sioncke, S. Van Elshocht, T. Verbiest, M. Kauranen, K. E. S. Phillips, T. J. Katz, and A. Persoons, "Circular-difference effects in second-harmonic generation from thin films," Synth. Met. 2001, 124, 191–193. 21. Karen E. S. Phillips, Thomas J. Katz, Steffen Jockusch, Andrew J. Lovinger, and Nicholas J. Turro, "Synthesis and Properties of an Aggregating Heterocyclic Helicene," J. Am. Chem. Soc. 2001, 123, 11899–11907. 22. Mikael Siltanen, Stefano Cattaneo, Elina Vuorimaa, Helge Lemmetyinen, Thomas J. Katz, Karen E. S. Phillips, and Martti Kauranen, "A regression technique to analyze the second-order nonlinear optical response of thin films," J. Chem. Phys. 2004, 121, 1–4. 23. Colin Nuckolls, Renfan Shao, Won-Gun Jang, Noel A. Clark, David M. Walba, and Thomas J. Katz, "Electrooptic Switching by Helicene Liquid Crystals," Chem. Mater. 2002, 14, 773–776. Back to Top  |
||

