Publications
Abstract for Society of Mining, Metallurgy and Exploration (SME) February 2007 meeting in Denver, CO.
Green Biosurfactants for Benign Mineral processing
P. Somasundaran 1 , Lei Zhang 3, Puspendu Deo 4, Jing Wang 1. and Richard A Gross 2, (1) Industry/University Cooperative Research Center for Surfactants, Columbia University, 500 W 120th Street, 910 S.W. Mudd, New York, NY 10027, Fax: 212-854-8362, [email protected], (2) NSF-I/UCRC Center for Biocatalysis and Bioprocessing of Macromolecules,Polytechnic University, (3) Akzo Nobel Chemicals, (4) CP Kelco
Biosurfactants formed by fermentation of renewable resources are “environmentally friendly” compounds that can modify mineral wettability suitably for use as collectors and modifiers. Colloidal and interfacial properties, such as aggregation, surface tension reduction and adsorption of natural sophorolipid surfactants, were investigated in this work to determine the role of alkyl moieties in formation of nanodomains on surfaces. Interestingly, these surfactants were found to adsorb selectively on certain substrates such as alumina but only weakly on others such as silica. We have also explored unique interfacial and colloidal properties of polysaccharides that are hydrophobically modified. Hydroxyethyl cellulose and its hydrophobically modified derivatives were studied systematically at talc-water interfaces using a combination of spectroscopic, microscopic, electrostatic adsorption and molecular modeling. The mechanism of talc-polysaccharide interactions was also explored. Modified interfacial and colloidal environments are indeed important in controlling adsorption and thereby flotation as well as dispersion/flocculation. Thus, solloidal (surface aggregate) structures formed by these compounds on solid substrates make them attractive candidates for use in mineral beneficiation as well as for pollution abatement techniques such as soil remediation and bioremediation.
Abstract for National Conference on Polymers, December 17-20, 2006 Pune, India
Effect of Modifying Functional Groups on Interfacial Behaviour of Hybrid Silicone Polymers
Somil Mehta,1 P. Somasundran,1
Columbia University, New York
Omprakash Yemul,2 Ravi Kulkarni2*
Elkay Chemicals Pvt Ltd, Pune, India.
Abstract
Polysiloxanes, are useful polymers, because of their high chemo-, thermo- stability and nontoxicity. The desired properties can be achieved by attaching suitable organic group to silicones. Such Organic/Inorganic hybrid silicone polymers are increasingly used in cosmetics; inks, paints, and fabric care applications due to their special Si - O bond characteristics. Though widely used, their utilization has mainly been empirical due to lack of fundamental knowledge. In most of the earlier studies the hydrophilic modification of silicones are based on non - ionic moieties. There is little information available about their ionic counterparts. The present work is focused on the effects of functionality on the conformation of chains at the air-water interface. The structures of functionally modified silicones have been presented in (Figure 1).
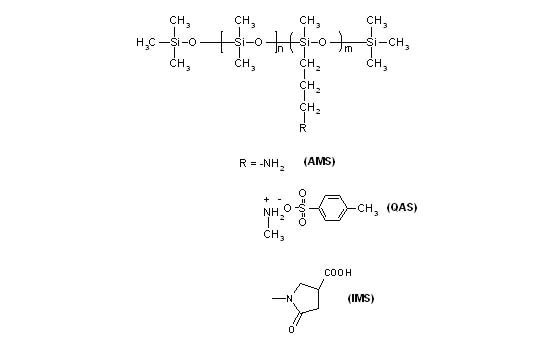
Figure 1 Structures of functionally modified silicones
Amino modified polymeric silicone (AMS) was synthesized from decamethyl cyclopentasiloxane (D5), tetramethyl ammonium hydroxide, and amino siloxane using equilibration process. The reactants were selected such that to have only one amino group per side chain. The ratio of reactants was adjusted such as to give m:n ratio of approximately 7.5:1 (figure 1) and the viscosity of the polymer was 65 cps. Quaternized amino silicone (QAS) was synthesized by methylation of the amine using methyl tosylate while acid modified silicone (IMS) has been carried out by reacting with itaconic acid (figure 1).
Hydrophilically grafted silicone polymers form monolayers at air-water interface, which is stabilized by interaction of functional groups that are submerged in water subphase. It was observed that the shape of the chain depends on the available area at the interface and there are conformational changes with increase in molecules per unit area. Unlike poly(dimethylsiloxane) (PDMS), which undergoes stretched-helix transition, hydrophilically grafted chains may form loops.
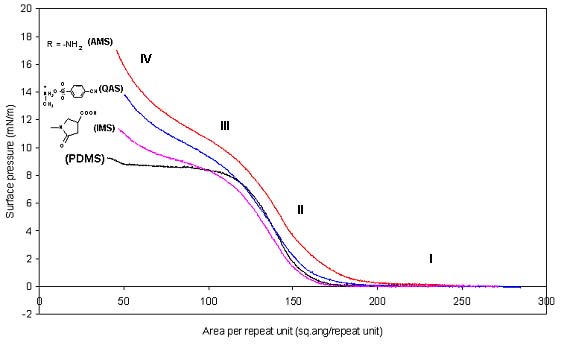
Figure 2 Surface pressure-area isotherms at air-water interface at 25 C for hydrophilically modified silicones compared with PDMS.
The superimposed isotherms of hydrophilically modified silicones and PDMS (figure 2) shows that the curves are almost the same upto 120Å2/repeat unit (i.e. in regions I and II), with slight differences possibly due to minor variations in the equivalent weights of respective polymers. This is proposed to be due to stretched conformation of chains in regions I and II, where only chain lengths play an important role and not the functional groups. On further compression, between 120Å2/repeat unit and 50Å2/repeat unit, the chain undergoes a transition from stretched conformation to helical or loop structure. In this process some of the functional groups are pulled or removed out of water. The functional group that interacts most strongly with water will offer maximum resistance to come out of water surface. Hence the chain having higher affinity for water undergoes higher compression and shows higher surface pressure. Based on this hypothesis, the slope of the curves in region III can be considered to be a measure of the hydrophilicity of the functional groups on the chain. A gradation in hydrophilicity of modified silicones at air-water interface has been predicted based on the langmuir isotherm and correlation with the scaling theory. According to scaling theory, the increasing gradation of affinity of functional groups for water is
AMS>QMS>IMS>PDMS. Similar trends have been reported in the past for end functionalized silicone chains.
(1) Hill, R.M., Silicone surfactants; Surfactant science series. Marcel Dekker: New York, 1996. 86.
(2) Hill, R.M., Silicone (siloxane) surfactants. In Encyclopedia of physical science and technology 3rd ed.;R. A. Meyers, Ed. Academic press: San Diego, CA 2002. 14: p. 793.
(3) E. D. Goddard, K.P.A., and P. Chandar, Silicone "superwetters". Langmuir, 1995. 11: p. 1415.
(4) Steven A. Snow, W.N.F., and Michael J. Owen, Synthesis and characterization of zwitterionic silicone sulfobetaine surfactants. Langmuir, 1990. 6: p. 385-391.
(5) Banks, W.H., Surface films of polydimethylsiloxanes on organic liquid substrates. Nature, 1954. 174: p. 365-366.
Abstract for SPE meeting, April 22-26, 2006 Tulsa OK
Paper proposal for the Fifteenth SPE Improved Oil Recovery Symposium
Mineral wettability control by optimizing adsorption of mixed surfactants
S. Lu and P. Somasundaran
Columbia University, New York
Abstract
Reagent adsorption on reservoir minerals is one of the most critical parameters in chemical flooding for improved oil recovery both due to their depletion and changes in mineral wettability leading to loss in sweep efficiency. The mechanisms that control both the extent of adsorption and the nature of the nanostructure of the adsorbed layer on relevant minerals were investigated in this work using mixtures of a series of surfactants of different charge and structure (nonionic alkyl ethoxylates, environmentally benign dodecyl-ß-D-maltoside (DM), anionic sodium dodecyl sulfonate and cationic single chain and Gemini amines) under various conditions of pH, salinity and, most importantly, mixing ratio of the surfactants. To elucidate the mechanisms involved, hydrophobicity of alumina particles was also monitored along with the charge of the particles and interfacial tension. It was discovered in this work that adsorption can be better controlled for maximum increase or reduction by using a mixture with one surfactant as an active adsorbate and the other as passive. For example while both DM and Sulfonate surfactants are active on alumina at pH 7 with maximum adsorption of both, interestingly only the Sulfonate component is active at pH 4 and the other component active at pH 10. It is proposed that the active species act as "anchors" for passive molecules to adsorb at pH10 due to hydrophobic chain-chain interaction but with the hydrophilic head groups oriented toward the bulk solution, making the mineral water wetted at lower bulk concentrations. On the basis of these results, it is proposed that active/passive systems with a low fraction of active component can be chosen to minimize adsorption and maximize the performance of surfactants in surfactant flooding.
Abstract for SPE meeting, April 22-26, 2006 Tulsa OK
Nano-structure of Mixed Surfactant Aggregates in Solution and on Minerals
R. Zhang and P. Somasundaran
Columbia University, New York
Abstract
Enhanced Oil Recovery techniques are aimed towards attracting the residual oil trapped in the reservoirs in the form of microdroplets between sand particles or inside porous rocks using, for example, by flooding with micellar solutions. Surfactants/polymers can change the wettability of the rock markedly and thus keep mobilizing the entrapped droplets. Since the nanostructures of the aggregates of surfactant mixtures determine particularly the orientation of the species, wettability as well as viscosity and the interfacial tension and thus the efficiency of the EOR process, it is useful to monitor the aggregates. The system of nonionic surfactant mixtures of n-dodecyl-ß-D-maltoside (DM) and nonyl phenol ethoxylated decyl ether (NP-10) were investigated in this work to indentify the role of the chemical structures of the surfactants in determining the natures of aggregates and the resultant surface modification of minerals. Using analytical ultracentrifuge technique coexistence of two types of micelles was discovered in solution of nonyl phenol ethoxylated decyl ether and its mixtures with n-dodecyl-ß-D-maltoside while only one micellar type was observed is present in n-dodecyl-ß-D-maltoside solutions. One type of micelles was spherical while the other was elongated.In the case of adsorption on minerals, interestingly NP-10 independently adsorbs on silica and DM only coadsorbs through hydrophobic chain chain interactions, while the role of each is reversed for the case of adsorption on alumina. For the case of mixtures, a modified packing parameter is proposed for predicting the formation of aggregates. The structures are spherical or cylindrical on silica but those on alumina undergo a spherical-to-cylindrical-to-bilayer transition with increasing concentration of the primary adsorbent. The maximum adsorption of the nonionic surfactant mixtures is determined by the size of hydrophilic groups of both surfactants and, importantly, the mixing ratio. This information enables tuning the formulations of the chemical floods for better sweep efficiency by varying parameters such as type of surfactants, molecular structures, mixing ratio as well as the total concentration.
3-D and side views of various nanostructures together with the V/Vfull ratio.
Abstract for SPE meeting, April 22-26, 2006 Tulsa OK
Conformational Behavior of Hydrophobically Modified Polymers and its Effect on the Stability of Emulsions and mineral wettability.
P. Somasundaran. T. Wines and P. Deo
Abstract
Hybrid polymers constitute a novel class of polymers that are finding important applications in rheological control, enhanced oil recovery, detergency, pharmaceuticals, paint and coating, etc, owing to their unique structural combination of both surfactants and polyelectrolytes. They form hydrophobic or hydrophilic nanodomains depending upon the environment in a manner similar to surfactants, but differing with respect to the size, shape, hydrophobicity/hydrophilicity of the internal core, etc. This unique property, together with the conformational sensitivity to pH, ionic strength and temperature, gives hybrids additional opportunities in rheological control, improved oil recovery and effluent treatment. In the current work, the solution and solid/liquid interfacial properties of poly
(maleic acid/octyl vinyl ether) (PMAOVE) has been investigated by studying hydrophobic aggregation, adsorption on solids and colloid stability using a multi pronged experimental approach. The flexible hydrophobic chains on
PMAOVE enable the polymer to form intra-molecular nanodomains in solutions as detected by pyrene fluorescence with the nature of the inside
nanodomain packing depending ,among other, on the solution pH.
Interestingly, the hypercoiled conformation of PMAOVE reduces its parking area at the alumina/water interface and thus shows a higher adsorption density than its unmodified homologue with the conformation sensitivity to pH also exhibited by its interfacial adsorption behavior. The effect of the hydrophobically modified polymer PMAOVE on the solution behavior of reverse microemulsions studied using electrical percolation, dynamic light scattering and electron spin resonance techniques made them useful for controlling the reverse microemulsion phase stability. These findings are expected to assist control of the solution and interfacial properties of flooding fluids and design of optimal additives in enhanced oil recovery schemes
Investigation of the Mobility of Amphiphilic Polymer - AOTReverse Microemulsion Systems Using Electron Spin Resonance
Thomas H. Wines; P. Somasundaran; Nichola J. Turro; Steffen
Jockusch; M. Francesca Ottaviani
NSF Industry/University Cooperative Research Center for Advanced Studies in Novel Surfactants, Columbia University, NewYork, NY, 10027, USA
Abstract
Amphiphilic polymers can be used as tools to manipulate the behavior of reverse microemulsions. EPR spectroscopy employing the spin probe 5-doxyl stearic acid was used to study the adsorption of a comb type polymer (polymaleic anhydride octyl vinyl ether) and a di-block polymer (polybutadiene-ethylene oxide) onto reverse microemulsion droplets formed from Aerosol-OT/ heptane/water. The findings indicate that the comb type polymer was adsorbed by the reverse microemulsion drops at low polymer concentrations causing a structural change of the micelle.
SPECTROSCOPIC INSIGHT INTO SURFACTANT-POLYMER INTERACTOINS IN SOLUTION AND AT SOLID LIQUID INTERFACES
P. Somasundaran
NSF Industry/University Cooperative Research Center for Advanced Studies in Novel Surfactants, Columbia University, NewYork, NY, 10027, USA
Abstract
Polymers and surfactants have applications in a variety of processes involving dispersion/flocculation, rheology control, deposition and coating, solubilization, encapsulation/release etc. In addition to adsorption density, the structure of the adsorbed layers determines the interfacial properties of particles in these systems. In this talk, the role of adsorbed layer nanostructural properties, namely nano-polarity, nano-fluidity and most importantly molecular conformation and orientation at solid-liquid interfaces in controlling the above interfacial processes will be discussed. A multi-pronged approach involving fluorescence, ESR, NMR, Resonance Raman and CAT Scan techniques, along with conventional techniques to measure turbidity, settling, surface charge and hydrophobicity was used to explore the adsorption mechanism and the structure of the adsorbed layers. The suspension stability and particles wettability display significant changes concomitant with changes in the structure of the adsorbed layer. When polymers are used, their conformation can be manipulated by changing solution conditions such as pH and/or by the addition of a second reagent. Such manipulation of polymer, particularly hybrid polymers can be used to obtain desired levels of stabilization or deposition of hydrophobic or hydrophilic particles.
The adsorption isotherms of surfactants on solids in aqueous show two to four different regions corresponding to different adsorption mechanisms. The colloidal properties also display significant changes concomitant with changes in the structure of the adsorbed layer. By changing the nature of solid surface, solution conditions or structure of the surfactant/polymer, adsorption as well as conformation of the adsorbed layer can be controlled. Such manipulation can lead to flocculation or dispersion even at the SAME adsorption density. Even the so called low molecular weight dispersants can act as good flocculants under appropriate dosage conditions. Controlling the adsorbed layers for desired performance will be discussed in this talk.
A bombardier beetle squirting benzoquinone poison or a "touch me not" plant folding up rapidly upon being attacked are examples of nanostructures in nature that provide inspiration for developing new classes of nanomaterials. Such materials can effectively meet the ever-increasing demand for reliable drug. In this talk, applications of these nanomaterials will be explored following a discussion of methods we have developed for producing and monitoring various nanostructures.
ROLE OF INTERACTIONS BETWEEN POLYMERS, SURFACTANTS AND PROTEINS IN MODIFYING SURFACES
P. Somasundaran
NSF Industry/University Cooperative Research Center for Advanced Studies in Novel Surfactants, Columbia University, NewYork, NY, 10027, USA
Abstract
A bombardier beetle squirting benzoquinone poison or a "touch me not" plant folding up rapidly upon being attacked or a lotus leaf rolling off water beads are examples of nanostructures in nature that provide inspiration for developing new classes of nanomaterials. In this talk, applications of such nanostructures for control of wettability, flocculation/dispersion, deposition, solubilization and biosurface modification will be explored following a discussion of methods developed for monitoring surfactant/polymer/protein aggregates in solutions and at solid/liquid interfaces. Techniques used include both spectroscopic techniques (fluorescence, ESR, NMR, Resonance Raman, surface plasmon resonance), advanced analytical (Analytical ultracentrifuge, ultrafiltration), scattering (neutron, light) techniques along with conventional techniques used to measure turbidity, surface charge and hydrophobicity.
In all cases, in addition to adsorption density, the nanostructure of the adsorbed layer determines the interfacial properties of particles in these systems. Thus depending on the orientation of the adsorbed species, flocculation or dispersion of suspensions can be obtained at the same polymer adsorption density and hydrophobicity or hydrophylicity of surfaces can be obtained at the same adsorption density of surfactant or hydrophobically modified polymers. In general while the wettability changes of particles correlate with hemimicellization of surfactants on them, stability of suspensions correlate with conformational changes of adsorbed polymers and proteins on surfaces. In this talk the role of nanostructure of adsorbed surfactant/polymer/protein layers in controlling various interfacial phenomena and techniques for obtaining desired colloidal performance will be discussed.
Controlled Dynamics of Poly(acrylic acid) Nanobrushes with Changes in pH, Ionic Strength and Time
Jing Wang, P. Somasundaran
NSF Industry/University Cooperative Research Center for Advanced Studies in Novel Surfactants, Columbia University, NewYork, NY, 10027, USA
Abstract
Poly(acrylic acid) nanobrushes were prepared on polystyrene-coated glass substrates via physisorption, and a number of techniques (Langmuir-Blodgett technique, X-ray photoelectron spectroscopy, water contact angle measurement, atomic force microscopy) were used to prepare and characterize the brushes. Contact angle measurements showed the polystyrene-coated glass surface to become more hydrophilic after poly(acrylic acid) grafting on it. XPS results confirmed the presence of PAA on the topmost surface of the substrate. From the results of AFM topographic imaging of the surface of PAA brush on PS-coated glass substrate, PAA brushes was found to form bigger and less densely distributed aggregates on the surface with a decrease in the grafting density. Also, PAA brush aggregates were found to undergo marked reversible conformational changes from coiled to stretched and vice-versa with changes in pH, ionic strength and contact time.
Complexations of poly(vinylcaprolactam) with anionic surfactants:
impact of surfactant molecular structures
Rui Zhang, Qiong Zhou, Larry Senak &
P. Somasundaran
Langmuir Center for Colloids and Interfaces
Columbia University, New York, N.Y. 10027
Abstract
Hydrophobically modified (HM) polymers with low consulate temperatures (LCST) have advantages over conventional polymers for certain applications as they display the behaviors of both surfactants and polymers. The poly(vinylcaprolactam) (PVCAP) has been characterized and its interactions with three anionic surfactants of various molecular structures, i.e., dodecyl sulfonate (C12SO3), sodium bis(2-ethylhexyl) sulfosuccinate (AOT) and disodium laureth 3 sulfosuccinate (SLE3) at air/solution interfaces and in aqueous solutions at neutral pH, have been studied in this work by measuring properties such as the surface activity, the phase behavior, and the binding cooperativity. At the air/aqueous solution interface, PVCAP participates in the monolayer formation of C12SO3 and AOT while no such behavior is observed for SLE3. Similarly in solution, PVCAP is found to interact with AOT and C12SO3, surprisingly, not with SLE3. The ion-dipole interaction is proposed to be the driving force to initiate the complexation for
the first two combinations at cac. Both the three EO groups and two charges in head group are responsible for the lack of reactivity for PVCAP-SLE3. At concentration above cac, the aggregation number of polymer-surfactant complex obtained from the binding isotherm follows the order of AOT < C12SO3 < SLE3. The results are interpreted with respect to surfactant molecular structures, such as the charge density and inorganic-organic balance. It is expected to tune the interaction of polymer and surfactant and obtain the desired performance.
Aggregate formation of binary nonionic surfactant mixtures on
hydrophilic surfaces
R. Zhang
Langmuir Center for Colloids and Interfaces
Columbia University, New York, N.Y. 10027
Abstract
Adsorption of surfactant mixtures on hydrophilic solid surfaces is of considerable theoretical and practical importance. Interestingly, in this study, cooperative adsorption of nonionic surfactant mixtures of nonyl phenol ethoxylated decyl ether (NP-10) and n-dodecyl-â-D-maltoside (DM) on silica and alumina has been investigated with a view to elucidate the nanostructures of their aggregates. In the mixed adsorption process, DM is identified to be the "active" component and NP is the "passive" one in the process of adsorption on alumina, while their roles are reversed on silica. Strong interaction between the surfactant molecules has been found for adsorption of surfactant mixtures on silica but weak effect is observed for the adsorption on alumina. The difference in the adsorptive interactions of the surfactants with the above minerals is attributed to the affinity of the active component to the solid surface and to the differences in the molecular structures of the surfactants. To better understand the interaction between surfactants at solid/solution interface from a molecular structure point of view, the nanostructures of mixed surface aggregates have been quantitatively predicted for the first time using a modified packing index. This work is expected to bringforth new sight to the understanding of structure-performance relationship.
Study of mixtures of n-dodecyl-ß-D-maltoside with anionic, cationic and nonionic surfactant in aqueous solutions using surface tension and fluorescence techniques
R. Zhang, L. Zhang, & P. Somasundaran
Langmuir Center for Colloids and Interfaces
Columbia University, New York, N.Y. 10027
Journal of Colloid and Interface Science, Volume 278, Issue 2, 15 October 2004, Pages 453-460
Abstract
Surfactants of practical interest are invariably mixtures of different types. In this study, mixtures of sugar-based n-dodecyl-ß-D-maltoside with cationic dodecyltrimethylammonium bromide, anionic sodium dodecylsulfate and nonionic pentaethyleneglycol monododecyl ether in solution, with and without supporting electrolyte, have been studied using surface tension and fluorescence spectroscopic techniques. Interaction parameters and mole fraction of components in mixed micelles were calculated using regular solution theory. The magnitude of interactions between n-dodecyl-ß-D-maltoside with other surfactants followed an order anionic/nonionic > cationic/nonionic > nonionic/nonionic mixtures. Since all surfactants have the same hydrophobic groups, strengths of interactions are attributed to the structures of hydrophilic headgroups. Electrolyte reduced synergism between n-dodecyl-ß-D-maltoside and ionic surfactant due to charge neutralization. Industrial sugar-based surfactant, dodecyl polyglucoside, yielded results similar to that by dodecyl maltoside, implying that tested commercial alkyl polyglucosides are similar to the pure laboratory samples in synergistic interactions with other surfactants. Fluorescence study not only supported the cmc results using tensiometry, but showed that interfaces of all the above mixed micelles/water are mildly hydrophobic. Based on these results, an attempt is made to discover the nature of interactions to be a combination of intermolecular potential energies, free energy due to packing of surfactant molecules in micelles.
Abnormal Micellar Growth in Sugar-based and Ethoxylated Nonionic Surfactants and Their mixtures in Dilute Regimes Using Analytical Ultracentrifugation
R. Zhang & P. Somasundaran
Langmuir Center for Colloids and Interfaces
Columbia University, New York, N.Y. 10027
Langmuir; (Article); 2004; ASAP Article; DOI: 10.1021/la049295a
Abstract
In order to develop structure-property relationships for surfactants that control their adsorption, solubilization and micellization behavior in mixed systems, and to develop predictive models based on such relationships, it is necessary to acquire quantitative information on various species present in these complex systems. Analytical ultracentrifugation technique is selected for the first time to characterize the species present in mixed micellar solutions due to its powerful ability to separate particles based on their size and shape. Two nonionic surfactants, n-dodecyl-ß-D-maltoside (DM) and nonyl phenol ethoxylated decyl ether (NP-10), and their 1:1 molar ratio mixtures were investigated in this study. Micelles of the nonionic surfactants and their mixtures are asymmetrical in shape at cmc. Interestingly, unlike ionic surfactants, the micellar growths of the nonionic surfactants were found to occur at concentration immediately above the cmc. The results from both sedimentation velocity and sedimentation equilibrium experiments suggest coexistence of two types of micelles in nonyl phenol ethoxylated decyl ether solutions and its mixtures with n-dodecyl-ß-D-maltoside while only one micellar species is present in n-dodecyl-ß-D-maltoside solutions. Type I micelles were primary micelles at cmc while type II micelles were elongated micelles. The differences in the micellar shapes of n-dodecyl-ß-D-maltoside and nonyl phenol ethoxylated decyl ether are attributed to packing parameters detected by their molecular structures.
Novel Nanogels for Encapsulation and Slow Release of Personal Care Actives
S. Chakraborty, P. Somasundaran & C. Gryte
NSF Industry/University Cooperative Center in Novel Surfactants
Columbia University, New York, NY 10025
Abstract
In personal care industries, it is a crucial problem to incorporate water-incompatible perfume molecules in reliable carriers and to release them at desired rates and desired sites. Nanogels are a type of spherical, covalently crosslinked polymeric networks with the particle size in nanometer range which can effectively encapsulate such materials. Since nanogels are small, porous and can swell/shrink in different environments and can be functionalized, they are potential carriers for fragrance and antimicrobial agents.
Nanogels were synthesized in this work by inverse microemulsion polymerization and by systematic modification, a series of functional nanogels, (hydrophobic nanogels, ionic nanogels and dually modified nanogels with both hydrophobic and ionic groups), were generated. Small molecule encapsulation experiments were carried out with these nanogels to determine the effects of functionalization. The nanogels chemically modified for hydrophobicity and electrostatic charge showed markedly higher ability for small molecule extraction as compared to the unmodified nanogels. In saline, although the efficiency of all types of nanogels was decreased, the hydrophobic nanogels showed much better performance than other types of nanogels. The range and capacity of the nanogels could be controlled by determining the effects of these functional groups on their loading properties. Mechanisms by which the functional groups influence the small molecule extraction are explored.
ENVIRONMENTALLY BENIGN SURFACTANTS FOR EFFICIENT ENHANCED OIL RECOVERYP.
Somasundaran and Q. Zhou
National Science Foundation I/UCR Center for Novel Surfactants
Langmuir Center for Colloids and Interfaces
Columbia University, New York, NY
Abstract
A large amount of oil is trapped in reservoirs made up of porous rocks after the traditional oil production. Various chemical methods have been under development to recover this oil, but they have been less than satisfactory due to the high cost of chemicals as well their loss by adsorption on reservoir rocks and precipitation. Indeed potential contamination of the environment by the chemicals is also a new important element to be considered. Thus there is a dire need to develop cost effective efficient environmentally benign chemicals to increase the recovery from oil reservoirs.
New surfactants capable of tolerating harsh conditions created by extremes of pH, temperature or inorganics and capable of interacting favorably with the polymers are promising in this regard. Such surfactants as sugar based ones and pyrrolidones are attracting attention, as they are potentially biodegradable. In many cases, mixed surfactants perform much better than single surfactants due to synergetic effects and ability to alleviate precipitation. Importantly, the effect of surfactants depend not only how much is adsorbed but also how they adsorb. A water wetted rock surface that is beneficial for displacement of oil can be obtained by manipulating the orientation of the adsorbed layers. Availability of modern equipment such as analytical ultracentrifuge, fluorescence and electron spin resonance spectrophotometers offers an unprecedented opportunity for elucidating the mechanisms involved and for designing optimum processes.
In this paper, problems and strategies for environmentally benign schemes for surfactants and polymers for improved oil recovery are considered. Some new schemes for coal cleaning based on the same principles are also briefly mentioned. Finally, opportunities for collaboration between the United States and Chinese researchers using their synthesis and analytical capabilities are indicated.
An EPR Investigation of the Adsorption of Dendrimers on Porous Surfaces
M. Francesca Ottaviani, Nicholas J. Turro,
Steffen Jockusch, Donald A. Tomalia
Abstract
Guest-host interactions of poly(amidoamine) dendrimers and porous surfaces, such as homoporous silica of different pore sizes, activated alumina (acidic, neutral, basic), and MCM-41 zeolite (Si/Al=30) were investigated by electron paramagnetic resonance (EPR) spectroscopy. Two generations of dendrimers were selected, G2 and G6, representing “early” and“late” generations, which were labeled with a nitroxide radical. It was found that the main parameters regulating the adsorption and interaction of the dendrimers and porous surfaces are: (I) the size of the pores of the porous materials (host) relative to the size of the dendrimer (guest), (II) the total surface area of the host that is accessible to the guest dendrimers, and (III) the chemical composition of the host and guest surfaces that come into contact upon adsorption. If the pore size of the host surface is smaller than the guest dendrimer diameter, then adsorption of the dendrimer occurs exclusively on the external host surface. If the pore size of the host surface is bigger than the dendrimer diameter, then the internal surface area is accessible to the dendrimers and large amounts of dendrimers are adsorbed. The chemical composition of the host surface and guest surface determines the strength of the host-guest interaction. An increase in alumina content (silica < zeolite MCM-41 < alumina) of the host increased the electrostatic interaction of the dendrimer with the host surface. Increased protonation of the external dendrimer amino groups also increased the electrostatic interaction. For the large dendrimers (G6), which possess a closed surface structure, electrostatic interactions are the main forces of host-guest interaction. Therefore, the strongest interaction of G6 was observed with basic alumina. For small dendrimers (G2), which possess an open structure, hydrophobic interactions between the low polarity internal dendrimer structure and the low polarity Si-O-Si groups of the host surface provide an additional host-guest interaction. High protonation of the dendrimer surface and high alumina content of the host surface inhibits this hydrophobic type of interactions. The combination of low polar Si-O-Si groups and highly polar AlO- groups in the zeolite MCM-41 probably cause the strongest interaction of the guest G2 dendrimer with MCM-41 compared to the other investigated host surfaces.
Polymer Adsorption and Conformation inDispersion/Flocculation of Concentrated Suspensions
Zhonghua Pan, P. Somasundaran
NSF Industry/University Cooperative Research Center for Advanced Studies in Novel Surfactants, Langmuir Center for Colloids and Interfaces
Columbia University, New York, N Y 10027
Abstract
Performance of polymers as stabilizers or flocculants for controlling the properties of concentrated suspensions depends upon the extent of the polymer adsorption as well as conformation at solid-liquid interfaces. Solids loading, polymer molecular weight and fractionation, and dissolved solid species, are important factors that control the polymer adsorption and conformation and the system behavior. In this study, polymer molecular weight fractionation and adsorption changes were monitored using stepwise adsorption tests and polydispersed polymer samples with techniques such as TOC (total organic carbon) and GPC (gel-permeation chromatography). Fluorescence and ESR (electron spin resonance) spectroscopy were used to explore the conformation/orientation of the polymers adsorbed on solid. Aluminum concentration of the residual solutions was determined using ICP (inductively coupled plasma) atomic emission. To investigate effects of dissolved alumina species on the system behavior, zeta potential measurements of the suspensions were made using a Zeta-Meter Model D.
It was found that the adsorption of PAA on alumina did not change significantly with increase in solids loading from 2 vol. % to 15 vol. %. Our earlier studies showed a “greater”adsorption density at low solids loading and a marked decrease in adsorption with increase in solids loading in the same range. Interestingly, it was found that the polymer adsorption behavior at high solids loading is different from that at low solids loading when the polymer sample contains impurity such as dioxane. Under the test conditions, smaller polymer molecules were found to preferentially adsorb first at the interfaces. Polymer molecular weight fractionation due to such preferential adsorption is more evident at low solids loading than at high solids loading, suggesting an increasing effect of particle-particle interaction on polymer diffusion. Concentration of the dissolved alumina species in the residual solutions was found to increase significantly as the system becomes denser, and this in turn affects the conformation of PAA in the solution: PAA molecules become more coiled in the presence of the dissolved alumina species at pH<7 even though the concentration of the species is low. Zeta potential of PAA-alumina suspensions with addition of dissolved alumina species was found to be markedly higher than that of PAA-alumina systems in the range of pH 4 to pH 10, suggesting that alumina surface becomes more positive in this pH range possibly due to increasing complexation of PAA with the dissolved alumina species.
Preliminary results of fluorescence and ESR spectra in our earlier studies suggested that the adsorbed PAA molecules tend to stretch out or dangle more into the solution as the system becomes denser. It is very clear that monodispersed polymer samples with fluorescence and spin labels are required to monitor the polymer conformation. More control experiments are also needed to monitor and minimize the effects of the labels on the system behavior.
Polyacrylic acid adsorption and conformation in concentrated alumina suspensions
Zhonghua Pan, Adrian Campbell, P. Somasundaran
NSF Industry/University Cooperative Research for Advanced Studies in Novel Surfactants
Langmuir Center for Colloids and Interfaces
Columbia University, New York, 10027, U.S.A.
Abstract
Particulate suspensions used in many industrial processes are typically highly concentrated and it is often difficult to keep them well dispersed or flocculated. Polymer adsorption is usually employed to achieve desired colloidal and rheological properties of such suspensions. In this work, adsorption of polyacrylic acid (PAA) in concentrated alumina dispersion has been monitored to determine the effect of solids loading on both the adsorption as well as the conformation of the adsorbed species. Fluorescence and electron spin resonance (ESR) spectroscopy were used to determine the conformation of labeled PAA adsorbed on alumina particles. Solids loading were found to have interesting effects on PAA adsorption and conformation at the alumina-water interface. As the solids loading is increased, the adsorption density of PAA was found to decrease under conditions of a fixed high initial polymer concentration. However, in the case of adsorption using reduced initial polymer concentration, this solid loading dependence was removed. Conformational studies (conducted at a constant adsorption density) showed the polymer molecules to become more stretched with increasing solids loading. The relationship between the polymer conformation and the particle dispersion was also investigated. Stretching of the adsorbed polymer was found in this case to produce an increase in the settling rate obtained, suggesting bridging effects.
Complexation between poly(maleic acid/octyl vinyl ether) and poly(vinyl caprolactam) in aqueous solution and at alumina/water interface
Q. Qiu and P. Somasundaran*
Langmuir Center for Colloids and Interfaces
Columbia University
520 West 120th Street, Room 911, New York, NY 10027
Abstract
Solution and interfacial properties of binary polymer mixtures of poly(maleic acid/octyl vinyl ether) (PMAOVE) and poly(vinyl caprolactam) (PVCAP) have been studied for the alumina/water system. To test the hydrophobic effect, mixtures of poly(maleic acid/methyl vinyl ether) (PMAMVE) and PVCAP is also investigated and compared to the behavior of PMAOVE/PVCAP. At low pH, both polymer mixtures become turbid upon mixing. The turbidity increases at low mixing ratios of PVCAP to the vinyl ether component, reaches a maximum and then decreases at higher mixing ratios. Upon shifting the pH to the alkaline range, i.e pH 7.5 and above, the turbid solution becomes clear for both the polymer mixtures. Cloud point measurements indicate absence of complexation of PVCAP with PMAMVE under the alkaline conditions, but strong interaction with PMAOVE. This is attributed to the different forces involved in the complexation among the polymers: H-bonding for PVCAP/PMAMVE, and both H-bonding and hydrophobic effects for PVCAP/PMAOVE. At alumina/water interface, the normally non-adsorbing PVCAP is triggered to adsorb by PMAOVE, attributed to the hydrophobic complexation between the two. However, the adsorption of PVCAP shows a maximum as a function of the concentration of PMAOVE. At concentrations of PMAOVE above the onset of its own plateau adsorption, the amount of PVCAP triggered to adsorb is reduced possibly due to the polymer complex formation in solution.
Hydrophobic Complexation of Poly(vinyl caprolactam) With Sodium Dodecylsulfate and Dodecyltrimethylammonium Bromide in Solution
Q.Qiu, P. Somasundaran, and B.A. Pethica
Langmuir Center for Colloids and Interfaces
Columbia University
500 West 120th Street, Room 911, New York, NY 10027
Abstract
The interaction of poly(vinyl caprolactam) (PVCAP) with sodium dodecylsulfate (SDS) and dodecyltrimethylammonium bromide (DTAB) in aqueous solutions has been studied systematically by measuring the phase separation temperature, hydrodynamic radius, pyrene solubility, and surfactant binding isotherms. Both surfactants were observed to elevate the solution cloud point of PVCAP and cause the polymer to undergo a coil to globule transition. This transition occurs at a concentration about a tenth of the critical micelle concentration (cmc) for SDS in the absence of polymer, but at the cmc with DTAB. The results indicate that PVCAP interacts with SDS monomers, but only with micelles in the case of DTAB. The phase behavior of the PVCAP/SDS/DTAB ternary system shows that the binding of SDS to PVCAP is reversible on changing the concentration of the free surfactant monomer in solution. Potentiometric titration of PVCAP solution and pyrene solubility in its mixtures with surfactants suggest that complexation of PVCAP and SDS is due to a combination of ion-dipole and hydrophobic effects. PVCAP and DTAB micelles interact through hydrophobic inclusion of polymer segments into the DTAB micelles. Pyrene is not solubilized by PVCAP in solution alone. Addition of SDS to PVCAP solutions induces marked pyrene solubilization well below the cmc, characterized by a region indicating saturation adsorption of the pyrene to the PVCAP/SDS complex. Above the SDS cmc solubilization of pyrene increases linearly with SDS concentration, corresponding to inclusion of the pyrene into the SDS micelles. In contrast to SDS, the addition of DTAB to a solution of PVCAP shows no pyrene solubilization until the cmc is reached.
Removal of Arsenic Species by the flotation of Ions
Zhonghua Pan, Lei Zhang and P. Somasundaran
NSF Industry/University Cooperative Research Center for Advanced Studies in Novel Surfactants, Langmuir Center for Colloids and Interfaces, Columbia University, New York, NY 10027, U.S.A.
Abstract
Foam Flotation, a relatively inexpensive technique, has been employed for the first time for enhanced removal of arsenic species from the solution. This method involves interactions between arsenic species and surfactants and removal of hydrophobic complexes formed by air bubbles under quiescent conditions. The system in our experiments consists of Arsenic (V) oxide hydrate, dodecylamine (CH3(CH2)11NH2), pyrogallol (C6H3(OH)3), or sodium dodecyl sulfate (CH3(CH2)11OSO3Na) aluminum chloride, and others. It was found that as much as 98% removal of arsenic species can be obtained, the removal being dependent upon the solution pH and the interactions between As species and surfactants used as activators or collectors. These results suggest that foam flotation based on the interactions between As species and surfactants is a new effective method either by itself or in combination with other techniques for the removal of As species from the solution.
International Synopisum of Arsenic in Drinking Water
November 26-27, Columbia University
Cyclic Voltammetric Study of Redox Reactions of Arsenic
Z.Wei, P. Duby, P. Somasundaran
Many treatment technologies are capable of removing arsenic from potable water. So far, adsorption of Arsenic (III and IV) by oxides of Aluminum, Iron and their mixtures has shown encouraging results. The present study is to provide the fundamental knowledge of the oxidation or reduction between arsenic species in water for further development of treatment techniques.
The redox reactions between As(III) and As(V) in acidic solution were investigated using cyclic voltammetric technique with a Pt rotating disk electrode (RDE). In the potential region corresponding to the reduction or oxidation of arsenic species only one cathodic and one anodic peak were observed, indicating that the redox reactions between As(III) and As(V) are controlled by one slow reaction, although two electrons are transferred. Judged by the shift of the peak potential when changing scan rate and the distance between anodic peak potential and cathodic peak potential, the redox reactions between As(III) and As(V) are irreversible slow reactions. Digital simulation will be employed to obtain kinetic information of the reactions.
Physicochemical Interactions In Arsenic Remediation: A Study Using Atomic Force Microscopy
D. Sarkar and P. Somasundaran
Abstract
Many treatment technologies are capable of removing arsenic from potable water, but the socio-economic situation of the affected region complicates the choice of treatment technologies. So far, adsorption of Arsenic (III and IV) by oxides of aluminum, iron and their mixtures has shown encouraging results. We are working to understand the basic interaction mechanism of arsenic species with such surfaces, using atomic force microscopy.
We investigated the deposition of arsenic species on glass surfaces, activated by ferric ions, after activation by ferric oxide particles failed to provide any evidence of arsenic deposition. With background knowledge that iron activates oxide minerals for adsorption of surfactant only in the pH window of 2 to 4, a monolayer adsorption of iron was attempted at pH 3.50. Such monolayer adsorption made it possible to study the interfacial processes by AFM. Initial results, does show deposition of the arsenic species on the glass surface, as sharp jagged precipitates.
Overcoming Contamination in Surface Plasmon Resonance Spectroscopy
D. Sarkar
Abstract
Surface plasmon resonance spectroscopy is a technique used for detection of subtle changes in the optical properties of materials, and finds wide application in biosensors and chemical tranducers. Due to the extreme sensitivity, contamination of the sensor surface is a major problem in experiments involving surface plasmon resonance spectroscopy. We report here that aluminum oxide (alumina) can be used as a perfect protective coating for the metal in surface plasmon resonance experiments. The alumina is electron beam evaporated to create a relatively thick layer on the sensor (gold) surface, immediately after the gold deposition process without exposing the latter to environmental conditions. During experiment, this protective coating was removed by dissolving the alumina in an alkaline solution, thus exposing the nascent, un-contaminated gold surface for further experiments. By studying the shift in the waveguide modes supported in the dielectric alumina layer, the dissolution of alumina was monitored. Theoretical simulation studies using Fresnel’s equations were done to explain the dissolution plots obtained. Atomic force microscopy of the alumina surface provided information on the variation of the roughness parameter, with the progress in dissolution. It was found that the roughness of the surface at all points during the dissolution process remained much below the wavelength of the incident radiation, thus justifying the modeling of the system as stacked Fresnel’s layers.
Conformational Dynamics of Polyacrylic Acid;
A Study Using Surface Plasmon Resonance Spectroscopy
D. Sarkar
Abstract
The conformational dynamics of polyacrylic acid induced by pH change is reported here. Polyacrylic acid immobilized on gold surface was exposed to pH changes and the conformational changes thus induced were followed in real time using surface plasmon resonance spectroscopy. The temporal profile of the stretching-coiling phenomenon showed an inflexion point, which was proposed to be arising due to the contradictory behavior of two different property changes in the polymeric system. Normally SPR response would be a convoluted effect of the thickness and refractive index changes, but the behavior observed here where the SPR response is predominantly governed by either one of the two, is unique and to the author’s knowledge is a feature, which is observed for the first time. Analysis of the kinetics of the angle change revealed that it takes longer for the polymer to stretch than it takes for it to collapse, with the kinetic rate constants varying by at least an order of magnitude. The SPR angle change as well as the kinetic constants increased linearly with molecular weight. Effect of Ca2+ was studied and it was found that the polymer was locked in its conformation due to the binding of this multivalent cations.
Polymer Surfactant Kinetics Using Surface Plasmon Resonance Spectroscopy Dodecyltrimethylammonium
Chloride / Polyacrylic Acid System
D. Sarkar
Abstract
Kinetics of polymer surfactant interactions and the effect of surfactant binding on the conformational dynamics of the polymer were explored in this work using surface plasmon resonance spectroscopy. Polyacrylic acid (PAA) was modified with thiol to varying degrees so as to force the polymer to form different loop sizes upon adsorption on the gold SPR sensor surface. Dodecyltrimethylammonium Chloride (DTAC) in solution was flowed over the polymer coated sensor surface and the binding was followed in real time. It was found that control of the loop size of the polymer on the solid surface enabled in turn the control of surfactant binding, with the largest loop allowing the maximum amount of surfactant to bind and vice versa. The kinetic plot of the binding showed three distinct segments. The first segment followed convective-diffusive kinetics. The second and third segments followed first order kinetics with the second rate being significantly faster than the first one. Careful analysis of the second segment showed that it is possible to divide it into two different segments, each following a first-order kinetics, with the second rate being slightly slower than the first one suggesting a gradual slow down of the reaction due to convolution from the polymer conformational changes. Mechanistically, the sudden increase in the rate for the third segment of surfactant binding implies that the polymer matrix is opening up so as to incorporate more surfactant molecules. This was attributed to the formation of charged double surfactant species the repulsive interaction in which prevented the polymer network from imploding. Studies using unmodified polymers suggested the possibility of sudden conformational rearrangement in the polymer network, with progress in surfactant binding. Furthermore, the reflectance of the SPR spectrum was found to increase upon surfactant binding implying that there is a decreased efficiency of coupling of the incident radiation into the surface plasmon mode of the metal, which suggested that the surfactant actually penetrated the polymer matrix.
Interfacial Dynamics of Macromolecules Using
Surface Plasmon Resonance Spectroscopy
D. Sarkar
Thesis Abstract
This thesis reports an in situ study of the conformational dynamics of polymers under external perturbation, using surface plasmon resonance spectroscopy. The research was undertaken to follow the response of stimulated macromolecules as the understanding of the basic interactions governing such, and the ability to gain control on the time evolution of three-dimensional structures is fundamental in developing smart materials of the future. SPR is a surface sensitive technique providing easy route to perturb a molecule and follow its relaxation in real time. As part of this research work a surface plasmon resonance spectroscope has been developed and the use of aluminum oxide (alumina) as a protective coating for the sensor surface has been reported for the first time.
The main aim of this project is to follow the conformational dynamics of macromolecules at solid/liquid interface. Polyacrylic acid (PAA) was chosen as the primary target molecule because of its chemistry and vast known literature. Response of immobilized PAA to changes in pH was followed in real time and the results correlated to the changing electrostatics of the system. The changing predominance of property changes governing the SPR response gave rise to an inflexion point, which to the author’s knowledge was observed for the first time. Interestingly, kinetic rate constants suggested that it takes longer for the polymer to stretch, than it takes for it to coil back, which was attributed to the decrease in local pH and the impermeability of the hydroxyl ions through the coiled polymer layer.
A second perturbation to the polyacrylic acid system was in the form of cationic surfactant (dodecyltrimethylammonium chloride) molecules. It was found that the amount of surfactant binding was directly proportional to the loop size of the polymer on the solid surface, which was controlled by modifying the polymer to varying degrees of thiolation. The temporal profile suggested the opening up of the polymer matrix due to the development of repulsive interactions, thus providing novel information on the conformational evolution of the polymer structure over time.


