Center Projects
Below is
a list of current Center projects and graduate students/post-docs
working on these projects:
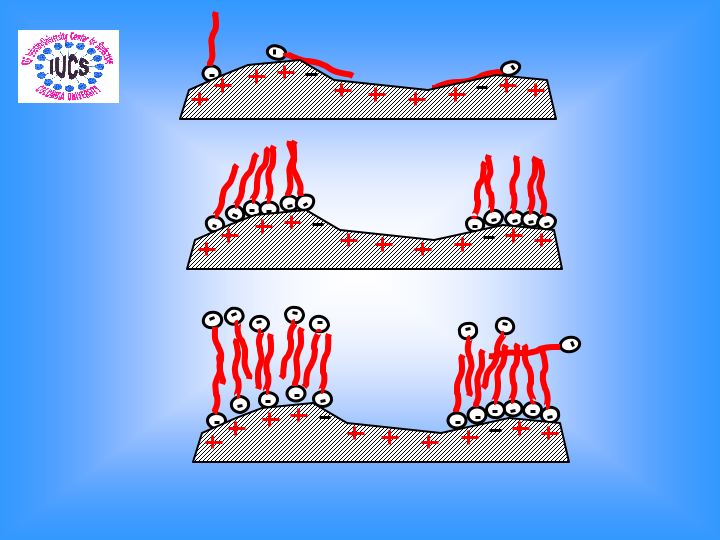
Novel polymeric nanoparticles for extraction and release of drugs and fragrance
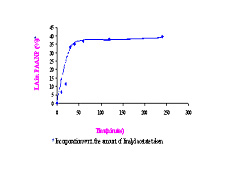
Nanoparticles are finding increasing applications as effective drug/attribute delivery devices. Previously, we have successfully designed modified poly(acrylamide) nanoparticles for overdose drug toxic extraction. The objective of the current project is to synthesize poly(acrylic acid) and other polymeric nanoparticles for incorporating fragrances and antimicrobial agents into the nanoparticles and study its subsequent release from the matrix. Novel polyacrylamide and poly(acrylic acid) nanoparticles (50-100 nm) have been synthesized by the reverse microemulsion method. The polyacrylamide nanoparticles after modification with charged and hydrophobic groups showed overdosed drug (amitriptyline) extraction of 80% compared to 18% for unmodified nanoparticles. The extraction of the fragrance, linalyl acetate by the nanoparticles we have recently studied. It was observed that 1% crosslinked poly(acrylic acid) nanoparticles could incorporate 38% of the linalyl acetate added to the system in 4hours. The efficacy of extraction increased when these nanoparticles were further modified with hydrophobic moieties like propyl amine and hexyl amine. Potential of poly(acryl amide) nanoparticles to extract vanillin by was also studied. When the release profile of incorporated linalyl acetate was monitored as a function of pH of the dispersion media, it was observed that the release was pH dependent. The potential of these nanoparticles for extraction of drugs like amitriptyline and bupivacaine is also being investigated. Surface Plasmon Resonance( SPR ) technique is being used to investigate short term extraction and release.
__________________________________________
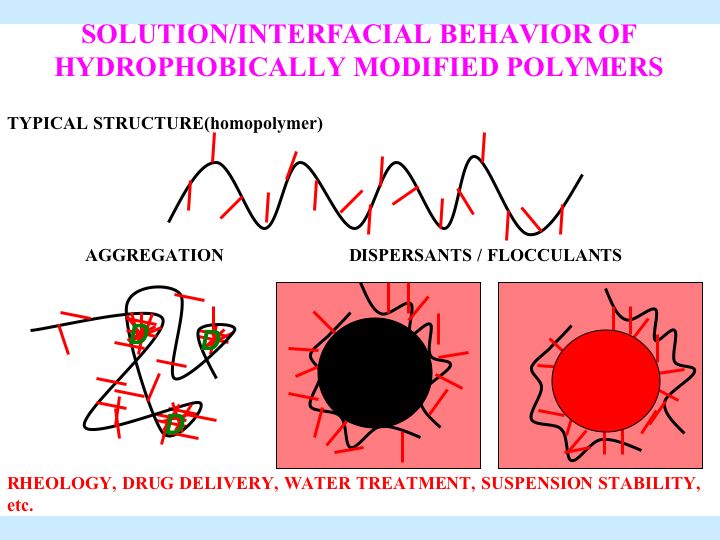
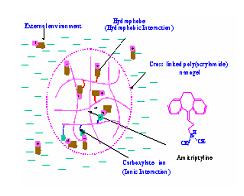
Solution and Interfacial Behavior of Hydrophobically Modified Polymers
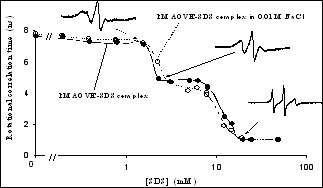
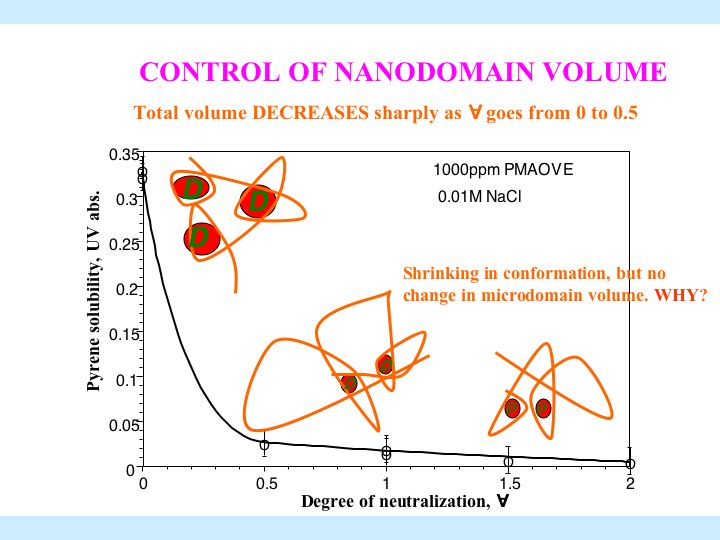
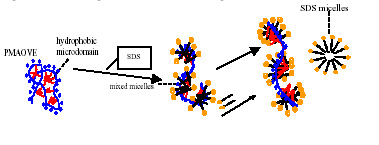
Hybrid modified polymers have features of both polymers and surfactants. Due to the associative nature of the hydrophobic groups, hybrid polymer can form intra-molecular as well as inter-molecular aggregates. This project aims to explore the adsorption, solubilization and turbidity properties of hydrophobically modified polymers in solutions along with their relevant colloidal applications. The association behavior, the major characteristic of hybrid polymers, is addressed with respect to their structure. Currently the interaction of several hybrid polymer surfactants such as poly(maleic acid/octyl viny ether) (PMAOVE) with sodium dodecyl sulfate ( SDS ) is studied. The ESR spectrum of 5-doxyl stearic acid (5-DSA), a probe molecule, indicates immobilization of the probe molecule in the hydrophobic nanodomains of the polymer in SDS -free solution. A sharp suggesting the structural reorientation of PMAOVE and formation of mixed micelles of PMAOVE and SDS ,. Upon further addition of SDS , the mobility of the probe remains constant, implying coexistence of SDS micelles and mixed micelles of PMAOVE and SDS . Such coexistence has major implications in their performances increase in the mobility of the probe was observed at around 2.2 mM and 8mM of SDS in colloidal processes.
________________________________________
Interfacial Behavior of Hydrophilically Modified Silicone surfactants
Siloxane based materials have become very important in industrial processes because of their unique properties in both aqueous and non-aqueous systems. Amongst them, a special class of hydrophilically modified molecules is termed silicone surfactants. These surfactants are commonly used as mold release agents, PU foam stabilizers, superspreaders for aqueous systems and emulsifiers in cosmetics.
We are investigating the interfacial characteristics of functionally grafted dimethyl siloxane backbone chain modified with various cationic, anionic and amphoteric functional groups.
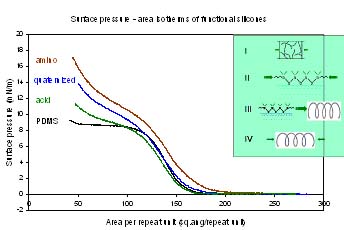
To study the effect of functional groups on the interfacial properties, surface pressure – area isotherms are measured at air – water interfaces. Various functional groups show a regular variation in slopes of the isotherm based on gradation in their hydrophilicity. The isotherms also show a transition in the conformation of the chains from stretched to helical or loops and trains. Using the above study, one therefore can identify what concentration of polymers is required in order to have a specific conformation of the chains, which in turn will help achieve desired properties properties.
__________________________________________
Modified Silicone polymers and their Interactions with Fabric Substrates
Our current project, which examines the interactions between silicones and fabrics, is aimed towards designing optimum surfactants and process schemes. Our primary objective is to establish a relationship between substrate properties and the particle size and surface charge of silicone emulsions as a function of their chemical structure that are used to treat them. So far we have used polydimethyl siloxane (PDMS) based amino silicone emulsions for our study. These silicones were quaternized to various degrees using dimethyl sulphate and then emulsified. The electrokinetic properties of these functional silicones are studied as a function of relevant conditions. It was expected that the silicone emulsions would show a steady positive zetapotential throughout the pH range due to quaternization by dimethyl sulphate. On the contrary, our results show a sudden drop in the zetapotential around pH 8 with the isoelectric point in the range of 9-10. The above results are important since they would help to better design the process conditions for the fabric treatments and also to design silicone based surfactants
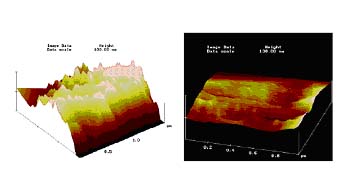
AFM was used as a tool to monitor the surface modification of fibers. It was observed that the treated fibers are far more smoothened, relaxed and uniform as compared to the untreated. Thus the morphology of the fabric is modified by treatment with specialty silicones. Furthermore, these results give a microscopic insight into the macroscopic properties such as softness and antiwrinkle of the silicone treated fabrics. We are using additional advanced analytical techniques such as the electron spin resonance spectroscopy and the fluorescence spectroscopy to study the system.
__________________________________________
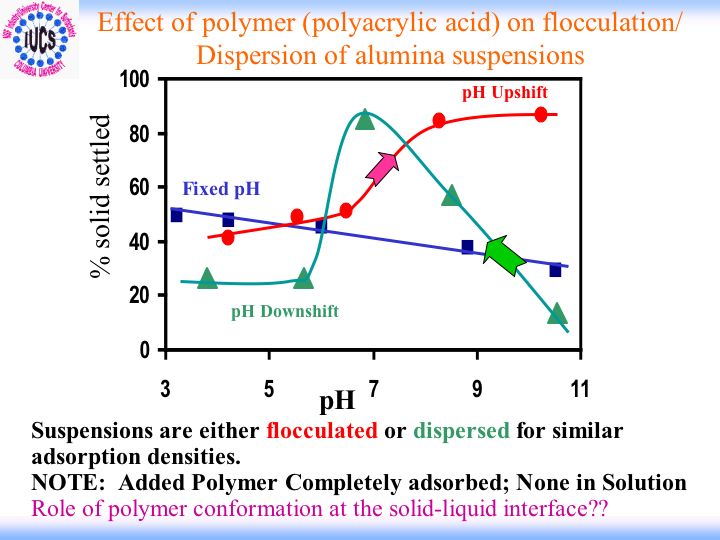
The Role of Surfactants and Polymers in Flocculation/Dispersion
Flocculation/dispersion of particles for proper processing and/or performance in industry is a critical and difficult issue to deal with, particularly when the solids content is rather high. It is our objective to understand the fundamentals involved in controlling the stability of concentratedsuspensions. Surfactants and polymers are added as stabilizers/flocculants in suchsystems. For polymers, the extent of adsorption and molecular conformation at the solid-liquid interface often determine their ability to perform properly. Foremost problem in this regard has been the difficulty in monitoring adsorption and particularly conformation in high solids loading suspensions.
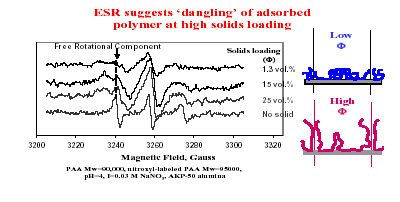
At the Center, in-situ monitoring of polymer adsorption and conformation in concentrated suspensions has been achieved by adapting the spectroscopic techniques such as fluorescence and ESR . We conducted a series of adsorption tests to check the adsorption behavior of polymers at different solid concentration in suspensions. It is noted that polymer conformation is changing with increasing solids loading. This is possibly due to preferential adsorption as a function of solids concentration.Effect of molecular weight distribution is therefore being investigated in detail using stepwise adsorption tests. The adsorption results from these tests along with chromatographic analysis of supernatants suggest preferential adsorption of polymers from a polydispersed system. These recent results imply preferential adsorption to play an important role in regulating the suspension stability and point out the significance of determining polymer adsorption and conformation. This in turn suggests possibilities for fine-tuning molecular weight distribution of additives in order to obtain desired dispersion characteristics.
__________________________________________
Mixed Surfactant Systems: Sugar-based surfactants in Mixtures
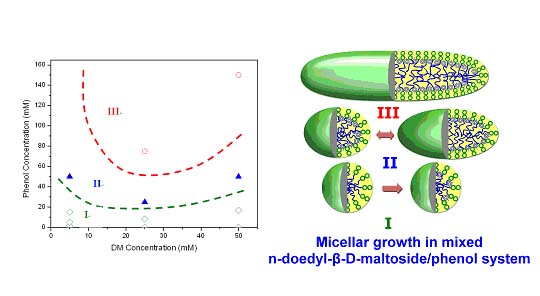
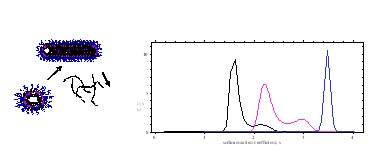
The project objective is to investigate interfacial phenomena such as adsorption, solubilization and micellization behavior of mixed surfactant systems and to explorethe synergistic effects of novel surfactant mixtures. Sugar-based surfactants are environmentally benign and have unique solution and interfacial properties. Gemini surfactants with two hydrophobic and two hydrophilic groups show more pronounced interfacial and solution properties. These surfactants have potential use in a wide variety of applications. The goal is to elucidate mechanisms governing solution and interfacial behavior of the mixed systems and the role of structural variations, and to generate a database for novel applications. Adsorption of nonionic / Gemini surfactant mixtures and nonionic / nonionic surfactant mixtures is studied. Analytical ultracentrifugation technique is employed for the first time to investigate the surfactant mixtures in solutions. Interestingly, unlike ionic surfactants, the micellar growths of DM and NP-10 surfactants and their mixtures were found to occur at concentrations immediately above the cmc. The results suggest coexistence of different types of micelles in NP-10 solutions and its mixtures with sugar-based surfactant while only one micellar species is present in sugar-based surfactant solutions. This technique is powerful for distinguishing the size and shape of various species in mixtures. Besides, both dynamic and equilibrium characteristics of microemulsion, nanoparticles, microgel for drug-delivery, polymer-surfactant and surfactant mixtures can be obtained using this technique.
__________________________________________
Study of the Adsorption and Conformation of Polysaccharides at Solid-Liquid Interface
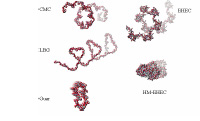
Polysaccharides have been widely used in many industrial sectors including those of foods, textiles, paper, adhesives, paints, pharmaceuticals, cosmetics and mineral processing. Their non-toxic and biodegradable properties make them ideal for industrial and personal care applications. Understanding of the interaction mechanisms between polysaccharides and solid surfaces can yield wider application of these polymers and development of novel synthetic polymers that can do an even better job. It is the aim of this work to explore the polysaccharide interactions and conformation at solid surfaces as a function of relevant parameters such as ionic strength, pH, temperature, solvent, polymer molecular weight and functional groups, solid composition, particle size etc. This work will focus on elucidating the mechanisms involved and obtain structure-performance relationships for interfacial behavior of polysaccharides with different structures. To achieve this goal, polysaccharides (carboxyl methyl cellulose, ethyl hydroxy ethyl cellulose (EHEC), hydrophobically modified EHEC, guar gum, locust bean gum etc.) with different functional groups are studied. A multipronged approach involving adsorption studies, electrophoretic mobility measurements, settling tests, fluorescence spectroscopy, FTIR- ATR (Raman), ESR , NMR, AFM and computer molecular simulation is used to investigate the mechanisms of interaction between these polymers and relevant substrates and derive a predictive model for polysaccharide structure-performance relationship. Computer simulation of molecular structure of: (a) CMC , (b) LBG, (c) Guar, (d) EHEC, (e) C 14-EHEC in aqueous environment yields information on their structure and properties.
__________________________________________
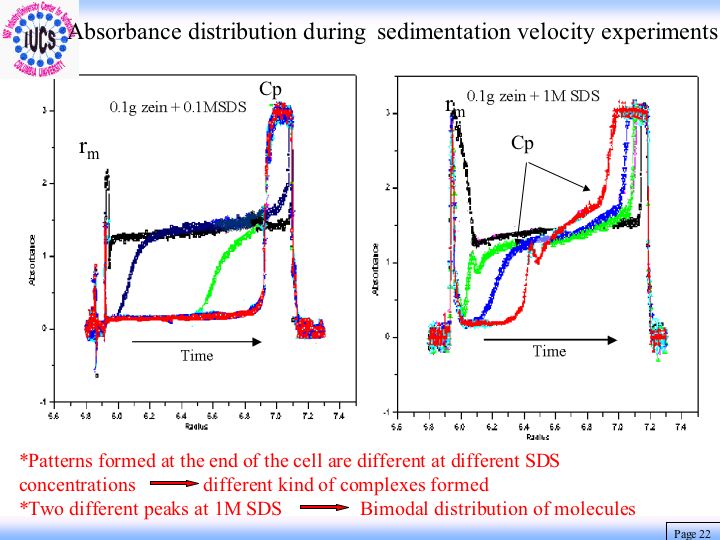
Interactions of Surfactants & Surfactant Mixtures with Model Membranes, Liposomes, Microbes and Antimicrobial Agents
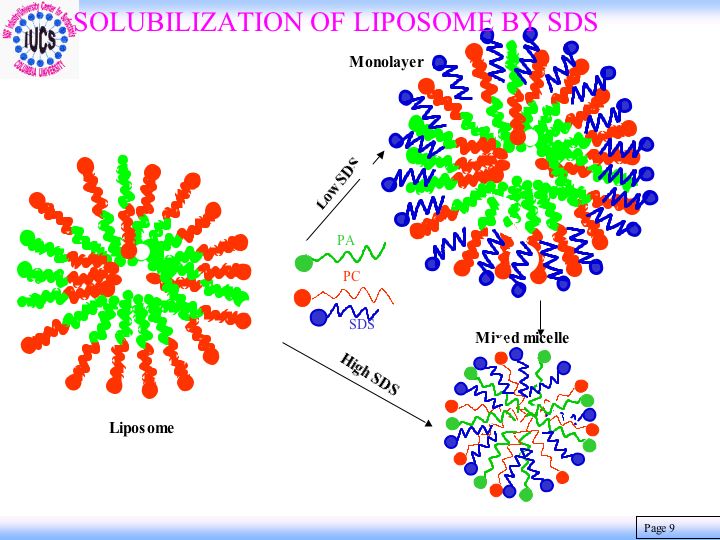
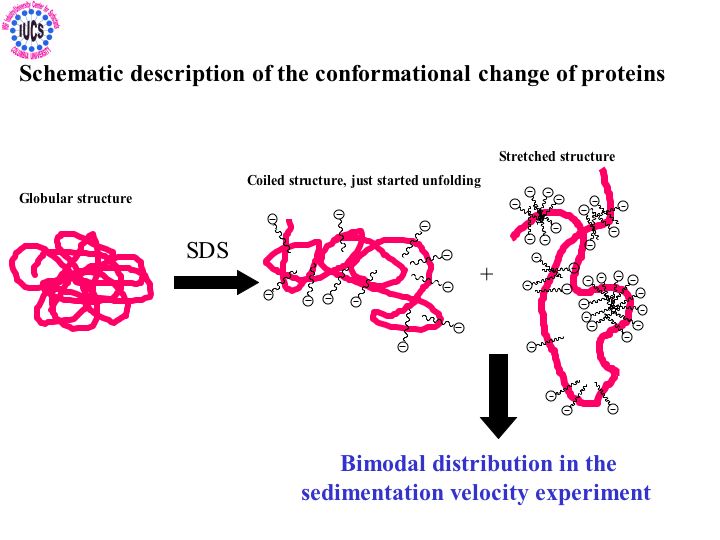
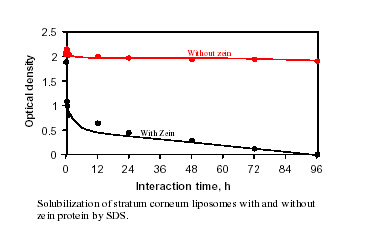
Surfactants are used as solubilizing and cleaning agents in the formulation of household and personal care products. For efficient and safe use of these surfactants, it is important to have knowledge about the mechanisms of their interactions with membrane proteins, lipids, and other components particular to human skin. That will enable us to develop a model and in turn to design the most suitable surfactant systems for specific applications. Information is being currently compiled for systems composed of surfactants, their mixtures, liposomes and proteins as well as ceramides that are components of such biomaterials as skins. Results of surface tension, ESR and fluorescence spectroscopy, viscosity, and mass spectroscopy showed solubilization of phosphatidic acid-phosphatidyl choline liposome by sodium dodecyl sulfate to be a process in which the phosphatidic acid exits first forming the mixed micelles and destabilizing the liposomes. It was discovered that while cholesterol stabilizes the liposomes, zein protein enhances their dissolution due to the binding of the SDS , unfolding and disrupting the regular packing of liposome components. Interestingly liposomes prepared with ceramide were found to be almost inert towards sodium dodecyl sulfate but again vulnerable in the presence of zein protein. Such infraction should prove useful for developing products that are effective but biologically benign.
__________________________________________
Study of Reagent Adsorption Kinetics Dynamics Using Surface Plasmon Resonance Spectroscopy
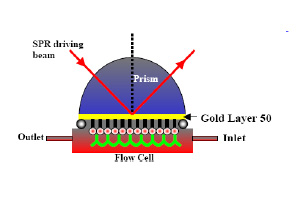
Specific reagent adsorption is the basis of many applications in industries such as microelectronics, mineral processing, cosmetics and personal care. Though considerable work has been done to understand the role of physico-chemical parameters of reagents and their adsorption, much less has been done to understand the kinetic aspects. In many processes the rate of the adsorption of the reagent is very important, for example in understanding the effects of various species on flotation behavior.
In our work we investigated the use of surface plasmon resonance spectroscopy ( SPR ) to study the dynamics of reagent adsorption on precious metals and their alloys on a short time scale (milliseconds).
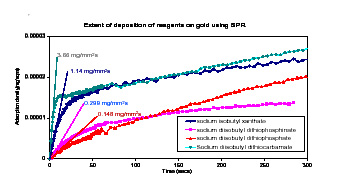
From SPR analysis it was observed that reagents with different functional groups have distinctively different binding rates. The rate of deposition of each reagent was calculated from theoretical considerations to obtain the adsorption density of reagent as a function of time. The limiting slope of this adsorption density vs. time graph at almost zero time was used as a tool to compare the relative rates of adsorption. A quantitative ranking was given to reagent adsorption kinetics based on the comparison of these limiting slopes. Such information will help the understanding of the real-life dynamic situations in flotation operation.
________________________________________
Aptamer Based Chemosensory Transducers, Toxic Scavengers or carrier Attributes
Nucleic acid aptamers are oligonucleotides of modest size (~15-100 nucleotides) that can bind to a particular ligand with great affinity and selectivity. Ligands can range from metal ions to small organic molecules to proteins to viruses and even to bacterial cells. Aptamers are created and selected using a combination of synthetic chemistry, enzymology and interfacial chemistry involving affinity chromatography. Oligonucleotides not only have the ability to bind specific ligands, but also in some cases can catalyze a chemical reaction involving the ligand. In these cases the ligand becomes a substrate. A few RNA-based enzymes of this sort (ribozymes) exist in nature; for the most part these exhibit RNA-cleaving activity. DNAzymes that cleave RNA or DNA at specific sequences have also been isolated through selection and amplification.
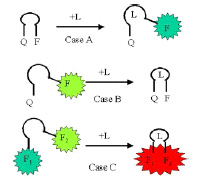
We propose here to develop aptamer-based biosensors in which ligand binding is coupled to the action of a protein enzyme to effect an amplification of several orders of magnitude. We start with a known DNAzyme with copper-dependent DNase activity; this DNAzyme is capable of self-cleavage at a specific resident single-stranded sequence. Amplification is effected by using this self-cleaving DNAzyme to tether a protein protease molecule to a solid support on one side of a small well. Each time a copper ligand binds to a tether, it self-cleaves, releasing the protease. Also present in the well is a short fluorescence probes connected to gold disks and embedded in a medium compatible with exposure to aqueous environments. The released protease diffuses to the gold disk and cleave the peptides releasing a fluorescent signal. _________________________________________
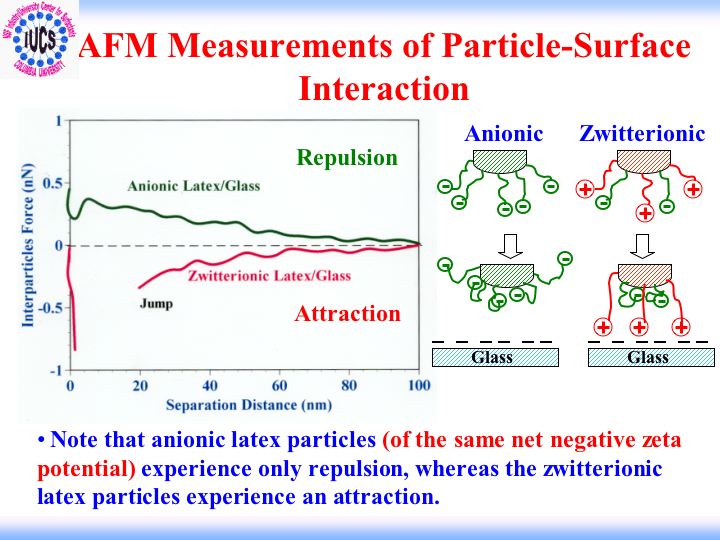
Adhesive Interactions between Particles in Aqueous and Non-Aqueous Media: Role of Surface Modification
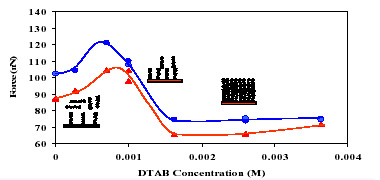
Adhesive force between particles is an important factor governing a number of phenomena involved in industrial processes. Direct measurement of that force can facilitate elucidation of mechanisms controlling adhesion as well as the identification of how to modify surface layers for optimum adhesion between particles, but there are no adequate techniques available for exploring the behavior of the typical particles present in real life industrial systems.
The objective of this project is to study molecular mechanisms of adhesive interactions between particles in dispersions. The study includes direct in-situ measurement of the adhesive forces between various particles and between particles and large surfaces. The behavior of adsorbed layers is being studied for different surfactant and polymer additives with particular emphasis on interaction time. Media effects are being examined to understand how different adsorbents modify contacts in different environments.
Finally, the general rules and guidelines for the designing of additives that will have the desired effects in any given system of particles and media will be developed.
_________________________________________
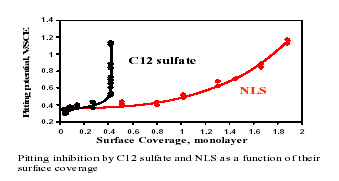
Inhibition of Pitting Corrosion by Surfactants
The main objective of this study is to investigate the mechanism by which organic surfactants act as pitting corrosion inhibitors and to identify key structural features responsible for such behavior. Inhibition of pitting corrosion of stainless steels in neutral saline solutions by surfactant and surfactant mixtures was studied during the current period and the inhibition mechanism was explored. The inhibition effect was determined using an electrochemical technique and correlated with the adsorption density of the surfactant on the stainless steel and the corresponding changes in surface charge. The current tests show that anionic N-lauroylsarcosine sodium salt (NLS) and anionic sodium dodecyl sulfate (C12 sulfate) are effective pitting inhibitors of AISI 304 stainless steel, while the cationic dodecyl tetramethyl ammonium chloride and the nonionic Triton X-100 are ineffective. The adsorption density of these surfactants on stainless steel and their effect on the surface charge of stainless steel were investigated using batch adsorption technique and electrophoretic mobility measurement, respectively. Based on the results, the mechanisms of pitting inhibition are explained in terms of the microstructure of the adsorbed surfactant layer and its effect on chloride ion adsorption. The inhibition mechanism will be further studied using contact angle and AFM techniques.
_________________________________________
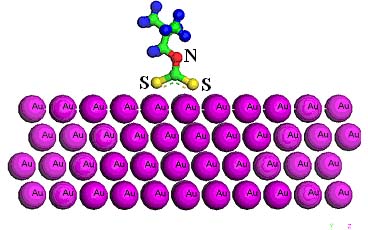
Structure-Property Correlation Studies Using Electronic Structure Calculations
Many new simulation techniques such as Dissipative particle dynamics (DPD), Coarse Grained Molecular Dynamics (CGMD), Stochastic Rotation Dynamics (SRD).., have been developed during the past few years for understanding and predicting molecular and mesoscale phenomena for systems such as polymers, colloids, and soft materials. We have initiated a program to utilize some of these simulation techniques to enhance our understanding of relevant problems such as adsorption, dispersion, and wetting etc. Aim of this program is two fold. First is to understand the dynamics at atomic/electronic levels, which is difficult to characterize using experiments with an acceptable degree of confidence. Second aim is to explain the experimental findings through a rational modeling framework, which are verifiable through subsequent simulations. For example, we have used ab-initio Density Functional Theory (DFT) calculation to simulate the electronic structure of various reagents that are currently used in precious metal extraction. The difference in charge distribution among the head groups area gave an idea on the reasons for differences in the reagent performance. Further, we have calculated parking area of both the head group and the chains of the optimized structures of these reagent molecules. This information along with the surface structure of the gold atom gives a good inference on reagent performance. We have now embarked on DFT simulation the relative bonding strength of these reagents on the gold surface. We believe that success in this approach will significantly reduce the time spent in developing new reagents for industries such as pharmaceuticals, health care, mineral processing etc,. Adsorption of sodium isobutyl dithiocarbamate on Au (111) surface
_________________________________________
Greener Biosurfactants and Their Interfacial Properties
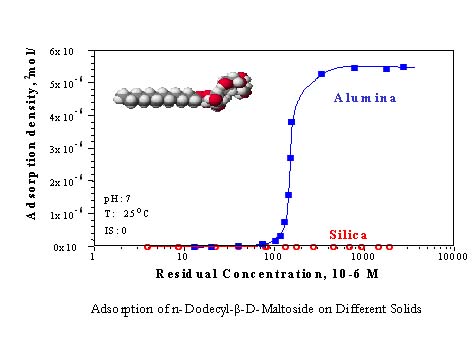
Biosurfactants are surface-active molecules synthesized by microorganisms grown on carbon sources such as hydrocarbons, sugars, vegetable and mineral oils. With the advantage of environmental compatibility, the demand for biosurfactants has been steadily increasing and may eventually replace their chemically synthesized counterparts. Biosurfactants have many advantages, such as biodegradability, low toxicity and environmental compatibility. For the sake of the environment, the use of biosurfactants is preferable to those of synthetic surfactants. According to their chemical composition and microbial origin, biosuractants are classified as 5 types: fatty acids, phospholipids, glycolipids, lipopeptides/lipoproteins and polymeric/particulate biosurfactants. Biosurfactants have amphiphatic properties since their complex structures are composed of hydrophilic and hydrophobic portions. As a consequence, biosurfactants can partition preferentially at the interfaces. Biosurfactants like sophrolipids, amygdalin-based amphiphiles, ascorbic acid-Based amphiphiles have been synthesized successfully. So far, the above biosurfactants can be modified to enhance their perfomance according to industrial requirements. Interfacial properties and solution behaviors such as n -dodecyl-β-D-maltoside have been investigated using techniques like surface tension, fluorescence spectroscopic, total organic carbon analysis techniques. It is our major aim to understand the unique behavior such as adsorption, as well as the structure-property-performance relationship of biosurfactants.
_________________________________________
A Study of Dispersion Phenomena Using Molecular Dynamics Simulations
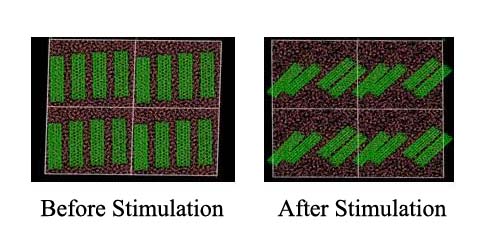
Aggregation of particles is a ubiquitous phenomenon in mineral and material processing industries. Reagents adsorbed onto the surfaces of these particles can be used to prevent aggregation and assist in particle dispersion. Dispersion has benefical applications in mineral processing, ceramics processing industries where the charge is kept dispersed (selectively or otherwise). In some cases, dispersion is unavoidable and detrimental, i.e. in the tar-sand industry; micro-emulsion of water-in-oil droplets laden with solid particles poses a major handicap in the oil-recovery and increase the cost of its production. When particle sizes reach nanometer levels their tendencies to aggregate are stronger. This problem is further complicated by the lack of clear quantitative methodology to estimate the degree of dispersion of these particles due to different reagents. Atomistic simulation techniques such as Molecular Dynamics (MD) offer an effective way of modeling and estimating the strength of reagent-particle interactions and their correlation with the dispersion. Consequently, two main objectives have been envisaged for this program. The first objective is to develop a correlation between dispersion and interaction energy of particle-reagent system. The second objective is to calculate transport properties such as viscosity, thermal conductivity etc..,. The above objectives will provide a direct means to evaluate and compare various reagents used in these studies through simple rheological experiments. A standard methodology for correlating the dispersion phenomena with interaction strength and reagent packing density would be established. Furhtermore, the study will aid in development of highly effective, novel dispersant for any given industrial condition, which can be tailored and developed at a very low cost. It is believed that the success in this program will significantly reduce the time spent in developing new reagents for industries such as ceramics processing, coating, polymer fillers, pharmaceuticals, health care, mineral processing etc,.
Aggregation observed in Carbon nanotube (CNT)-water suspensions
________________________________________
Adsorption of mixed surfactants at solid/liquid interface
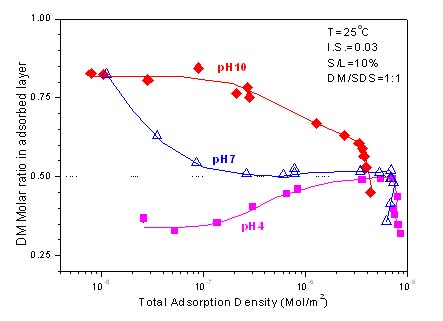
Surfactant aggregation at solid-liquid interfaces can result in unique nano domains with applications in processes, such as oil recovery, flotation and derpersion. In these applications, mixed surfactants are invariably used because of both the synergistic interactions among surfactants and the economic benefits. In this project, we studied the aggregation behavior of mixtures of environmentally benign sugar-based surfactants and other types of surfactants at solid/liquid interfaces as a function of relevant parameters such as pH, ionic strength and solid properties. Surfactant mixtures were studied using depletion technique, two-phase extraction hydrophobicity determination, surface tensiometry, static fluorescence spectroscopy and analytical ultracentrifugation to elucidate the mechanism of adsorption of mixtures, structure of aggregates at solid/liquid interface and micellization behavior. Adsorption of mixtures of n-Dodecyl- β -D-maltoside and sodium dodecyl sulfonate on alumina, as a function of mixing ratio, were measured at different pH. Interestingly, results obtained showed strong synergistic interactions at pH 4 and pH 10 and antagonism at pH 7. CMC values of surfactant mixtures determined by surface tension measurement were compared to those calculated by regular solution theory. A negative interaction parameter β showed moderate interaction between dodecyl maltoside and dodecyl sulfonate. Results obtained from fluorescence were in good agreement with those obtained by surface tension measurement.
________________________________________
Aggregation of mixed surfactants studied using analytical ultracentrifuge
The behavior of mixed surfactant is often determined by the nanostructures of aggregates in solutions in many applications. Also information on micellization of mixed surfactant helps understand the adsorption properties at solid/liquid interface. However, such information is often missing due to the lack of powerful techniques. Analytical ultracentrifuge was employed to elucidate surfactant micellar information in terms of aggregate number, micelle size and shape, etc. The experiments were run at 40000 rpm and 25.0 ℃ . The sedimentation velocity curves were scanned for 15 hours. Afterwards, the results were analyzed using software. By fitting the velocity curves, the distribution of sedimentation coefficient for mixed surfactants is obtained. In case of n-Dodecyl- β -D-maltoside and sodium dodecyl sulfonate, these two surfactants have very close specific volumes, but form very different micelles. Spherical micelles are identified in this mixture system. The shape and size of micelles suggest the possible nanostructure of aggregates formed at solid/liquid interface.